| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50193172 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_422157 (CHEMBL863594) |
|---|
| IC50 | 7000±n/a nM |
|---|
| Citation |  Lau, JF; Jeppesen, CB; Rimvall, K; Hohlweg, R Ureas with histamine H3-antagonist receptor activity--a new scaffold discovered by lead-hopping from cinnamic acid amides. Bioorg Med Chem Lett16:5303-8 (2006) [PubMed] Article Lau, JF; Jeppesen, CB; Rimvall, K; Hohlweg, R Ureas with histamine H3-antagonist receptor activity--a new scaffold discovered by lead-hopping from cinnamic acid amides. Bioorg Med Chem Lett16:5303-8 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50193172 |
|---|
| n/a |
|---|
| Name | BDBM50193172 |
|---|
| Synonyms: | (4-tert-butylpiperidin-1-yl)(4-isopropylpiperazin-1-yl)methanone | CHEMBL219128 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H33N3O |
|---|
| Mol. Mass. | 295.4634 |
|---|
| SMILES | CC(C)N1CCN(CC1)C(=O)N1CCC(CC1)C(C)(C)C |
|---|
| Structure | 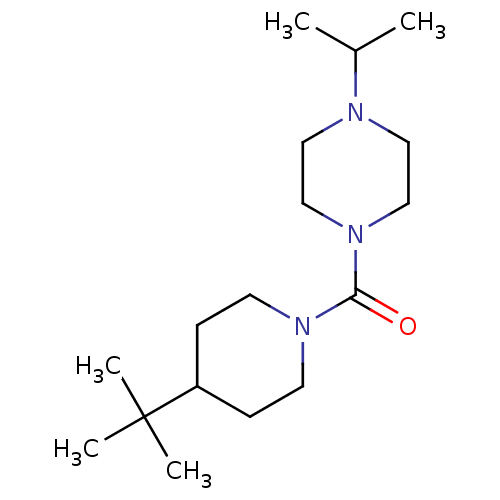
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lau, JF; Jeppesen, CB; Rimvall, K; Hohlweg, R Ureas with histamine H3-antagonist receptor activity--a new scaffold discovered by lead-hopping from cinnamic acid amides. Bioorg Med Chem Lett16:5303-8 (2006) [PubMed] Article
Lau, JF; Jeppesen, CB; Rimvall, K; Hohlweg, R Ureas with histamine H3-antagonist receptor activity--a new scaffold discovered by lead-hopping from cinnamic acid amides. Bioorg Med Chem Lett16:5303-8 (2006) [PubMed] Article