| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50193520 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_385312 (CHEMBL869229) |
|---|
| IC50 | 7600±n/a nM |
|---|
| Citation |  Deng, BL; Hartman, TL; Buckheit, RW; Pannecouque, C; De Clercq, E; Cushman, M Replacement of the metabolically labile methyl esters in the alkenyldiarylmethane series of non-nucleoside reverse transcriptase inhibitors with isoxazolone, isoxazole, oxazolone, or cyano substituents. J Med Chem49:5316-23 (2006) [PubMed] Article Deng, BL; Hartman, TL; Buckheit, RW; Pannecouque, C; De Clercq, E; Cushman, M Replacement of the metabolically labile methyl esters in the alkenyldiarylmethane series of non-nucleoside reverse transcriptase inhibitors with isoxazolone, isoxazole, oxazolone, or cyano substituents. J Med Chem49:5316-23 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50193520 |
|---|
| n/a |
|---|
| Name | BDBM50193520 |
|---|
| Synonyms: | (E)-5-[1-(3-methoxy-7-methyl-benzo[d]isoxazol-5-yl)-4-(2-oxooxazolidin-3-yl)but-1-enyl]-2,7-dimethylbenzo[d]isoxazol-3-one | CHEMBL387003 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H25N3O6 |
|---|
| Mol. Mass. | 463.4825 |
|---|
| SMILES | COc1noc2c(C)cc(cc12)C(=C/CCN1CCOC1=O)\c1cc(C)c2on(C)c(=O)c2c1 |
|---|
| Structure | 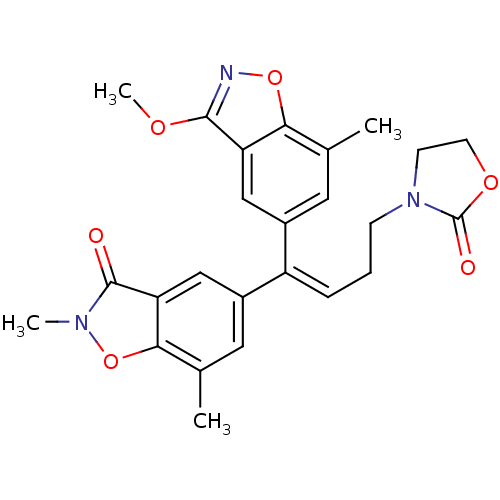
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Deng, BL; Hartman, TL; Buckheit, RW; Pannecouque, C; De Clercq, E; Cushman, M Replacement of the metabolically labile methyl esters in the alkenyldiarylmethane series of non-nucleoside reverse transcriptase inhibitors with isoxazolone, isoxazole, oxazolone, or cyano substituents. J Med Chem49:5316-23 (2006) [PubMed] Article
Deng, BL; Hartman, TL; Buckheit, RW; Pannecouque, C; De Clercq, E; Cushman, M Replacement of the metabolically labile methyl esters in the alkenyldiarylmethane series of non-nucleoside reverse transcriptase inhibitors with isoxazolone, isoxazole, oxazolone, or cyano substituents. J Med Chem49:5316-23 (2006) [PubMed] Article