| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase Fyn |
|---|
| Ligand | BDBM50210272 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_434554 (CHEMBL914201) |
|---|
| IC50 | 27300±n/a nM |
|---|
| Citation |  Dinges, J; Albert, DH; Arnold, LD; Ashworth, KL; Akritopoulou-Zanze, I; Bousquet, PF; Bouska, JJ; Cunha, GA; Davidsen, SK; Diaz, GJ; Djuric, SW; Gasiecki, AF; Gintant, GA; Gracias, VJ; Harris, CM; Houseman, KA; Hutchins, CW; Johnson, EF; Li, H; Marcotte, PA; Martin, RL; Michaelides, MR; Nyein, M; Sowin, TJ; Su, Z; Tapang, PH; Xia, Z; Zhang, HQ 1,4-Dihydroindeno[1,2-c]pyrazoles with acetylenic side chains as novel and potent multitargeted receptor tyrosine kinase inhibitors with low affinity for the hERG ion channel. J Med Chem50:2011-29 (2007) [PubMed] Article Dinges, J; Albert, DH; Arnold, LD; Ashworth, KL; Akritopoulou-Zanze, I; Bousquet, PF; Bouska, JJ; Cunha, GA; Davidsen, SK; Diaz, GJ; Djuric, SW; Gasiecki, AF; Gintant, GA; Gracias, VJ; Harris, CM; Houseman, KA; Hutchins, CW; Johnson, EF; Li, H; Marcotte, PA; Martin, RL; Michaelides, MR; Nyein, M; Sowin, TJ; Su, Z; Tapang, PH; Xia, Z; Zhang, HQ 1,4-Dihydroindeno[1,2-c]pyrazoles with acetylenic side chains as novel and potent multitargeted receptor tyrosine kinase inhibitors with low affinity for the hERG ion channel. J Med Chem50:2011-29 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase Fyn |
|---|
| Name: | Tyrosine-protein kinase Fyn |
|---|
| Synonyms: | FYN | FYN_HUMAN | Proto-oncogene Syn | Proto-oncogene c-Fyn | SLK | SRC | Src-like kinase | p59-Fyn |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 60762.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06241 |
|---|
| Residue: | 537 |
|---|
| Sequence: | MGCVQCKDKEATKLTEERDGSLNQSSGYRYGTDPTPQHYPSFGVTSIPNYNNFHAAGGQG
LTVFGGVNSSSHTGTLRTRGGTGVTLFVALYDYEARTEDDLSFHKGEKFQILNSSEGDWW
EARSLTTGETGYIPSNYVAPVDSIQAEEWYFGKLGRKDAERQLLSFGNPRGTFLIRESET
TKGAYSLSIRDWDDMKGDHVKHYKIRKLDNGGYYITTRAQFETLQQLVQHYSERAAGLCC
RLVVPCHKGMPRLTDLSVKTKDVWEIPRESLQLIKRLGNGQFGEVWMGTWNGNTKVAIKT
LKPGTMSPESFLEEAQIMKKLKHDKLVQLYAVVSEEPIYIVTEYMNKGSLLDFLKDGEGR
ALKLPNLVDMAAQVAAGMAYIERMNYIHRDLRSANILVGNGLICKIADFGLARLIEDNEY
TARQGAKFPIKWTAPEAALYGRFTIKSDVWSFGILLTELVTKGRVPYPGMNNREVLEQVE
RGYRMPCPQDCPISLHELMIHCWKKDPEERPTFEYLQSFLEDYFTATEPQYQPGENL
|
|
|
|---|
| BDBM50210272 |
|---|
| n/a |
|---|
| Name | BDBM50210272 |
|---|
| Synonyms: | 6-((4-methylpiperazin-1-yl)methyl)-3-(5-(3-phenoxyprop-1-ynyl)thiophen-3-yl)-1,4-dihydroindeno[1,2-c]pyrazole | CHEMBL390156 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H28N4OS |
|---|
| Mol. Mass. | 480.624 |
|---|
| SMILES | CN1CCN(Cc2ccc-3c(Cc4c-3n[nH]c4-c3csc(c3)C#CCOc3ccccc3)c2)CC1 |
|---|
| Structure | 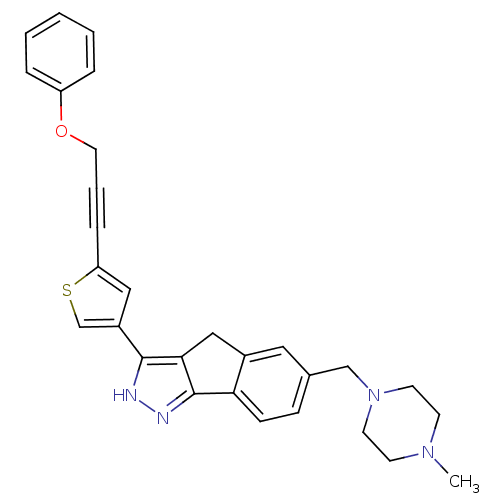
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dinges, J; Albert, DH; Arnold, LD; Ashworth, KL; Akritopoulou-Zanze, I; Bousquet, PF; Bouska, JJ; Cunha, GA; Davidsen, SK; Diaz, GJ; Djuric, SW; Gasiecki, AF; Gintant, GA; Gracias, VJ; Harris, CM; Houseman, KA; Hutchins, CW; Johnson, EF; Li, H; Marcotte, PA; Martin, RL; Michaelides, MR; Nyein, M; Sowin, TJ; Su, Z; Tapang, PH; Xia, Z; Zhang, HQ 1,4-Dihydroindeno[1,2-c]pyrazoles with acetylenic side chains as novel and potent multitargeted receptor tyrosine kinase inhibitors with low affinity for the hERG ion channel. J Med Chem50:2011-29 (2007) [PubMed] Article
Dinges, J; Albert, DH; Arnold, LD; Ashworth, KL; Akritopoulou-Zanze, I; Bousquet, PF; Bouska, JJ; Cunha, GA; Davidsen, SK; Diaz, GJ; Djuric, SW; Gasiecki, AF; Gintant, GA; Gracias, VJ; Harris, CM; Houseman, KA; Hutchins, CW; Johnson, EF; Li, H; Marcotte, PA; Martin, RL; Michaelides, MR; Nyein, M; Sowin, TJ; Su, Z; Tapang, PH; Xia, Z; Zhang, HQ 1,4-Dihydroindeno[1,2-c]pyrazoles with acetylenic side chains as novel and potent multitargeted receptor tyrosine kinase inhibitors with low affinity for the hERG ion channel. J Med Chem50:2011-29 (2007) [PubMed] Article