| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50272049 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_555862 (CHEMBL953406) |
|---|
| IC50 | 800±n/a nM |
|---|
| Citation |  Cai, ZW; Wei, D; Schroeder, GM; Cornelius, LA; Kim, K; Chen, XT; Schmidt, RJ; Williams, DK; Tokarski, JS; An, Y; Sack, JS; Manne, V; Kamath, A; Zhang, Y; Marathe, P; Hunt, JT; Lombardo, LJ; Fargnoli, J; Borzilleri, RM Discovery of orally active pyrrolopyridine- and aminopyridine-based Met kinase inhibitors. Bioorg Med Chem Lett18:3224-9 (2008) [PubMed] Article Cai, ZW; Wei, D; Schroeder, GM; Cornelius, LA; Kim, K; Chen, XT; Schmidt, RJ; Williams, DK; Tokarski, JS; An, Y; Sack, JS; Manne, V; Kamath, A; Zhang, Y; Marathe, P; Hunt, JT; Lombardo, LJ; Fargnoli, J; Borzilleri, RM Discovery of orally active pyrrolopyridine- and aminopyridine-based Met kinase inhibitors. Bioorg Med Chem Lett18:3224-9 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50272049 |
|---|
| n/a |
|---|
| Name | BDBM50272049 |
|---|
| Synonyms: | 1-(4-(2-amino-3-ethynylpyridin-4-yloxy)-3-fluorophenyl)-3-(2-(4-fluorophenyl)acetyl)urea | CHEMBL502531 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H16F2N4O3 |
|---|
| Mol. Mass. | 422.3842 |
|---|
| SMILES | Nc1nccc(Oc2ccc(NC(=O)NC(=O)Cc3ccc(F)cc3)cc2F)c1C#C |
|---|
| Structure | 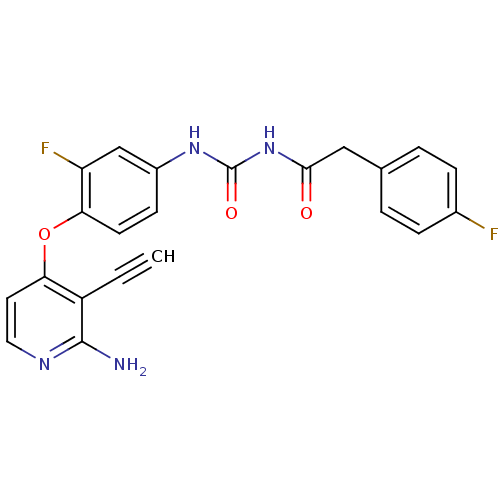
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cai, ZW; Wei, D; Schroeder, GM; Cornelius, LA; Kim, K; Chen, XT; Schmidt, RJ; Williams, DK; Tokarski, JS; An, Y; Sack, JS; Manne, V; Kamath, A; Zhang, Y; Marathe, P; Hunt, JT; Lombardo, LJ; Fargnoli, J; Borzilleri, RM Discovery of orally active pyrrolopyridine- and aminopyridine-based Met kinase inhibitors. Bioorg Med Chem Lett18:3224-9 (2008) [PubMed] Article
Cai, ZW; Wei, D; Schroeder, GM; Cornelius, LA; Kim, K; Chen, XT; Schmidt, RJ; Williams, DK; Tokarski, JS; An, Y; Sack, JS; Manne, V; Kamath, A; Zhang, Y; Marathe, P; Hunt, JT; Lombardo, LJ; Fargnoli, J; Borzilleri, RM Discovery of orally active pyrrolopyridine- and aminopyridine-based Met kinase inhibitors. Bioorg Med Chem Lett18:3224-9 (2008) [PubMed] Article