| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene A-4 hydrolase |
|---|
| Ligand | BDBM50251385 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_488137 (CHEMBL987428) |
|---|
| IC50 | 28±n/a nM |
|---|
| Citation |  Ye, B; Bauman, J; Chen, M; Davey, D; Khim, SK; King, B; Kirkland, T; Kochanny, M; Liang, A; Lentz, D; May, K; Mendoza, L; Phillips, G; Selchau, V; Schlyer, S; Tseng, JL; Wei, RG; Ye, H; Parkinson, J; Guilford, WJ Synthesis of N-alkyl glycine amides as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem Lett18:3891-4 (2008) [PubMed] Article Ye, B; Bauman, J; Chen, M; Davey, D; Khim, SK; King, B; Kirkland, T; Kochanny, M; Liang, A; Lentz, D; May, K; Mendoza, L; Phillips, G; Selchau, V; Schlyer, S; Tseng, JL; Wei, RG; Ye, H; Parkinson, J; Guilford, WJ Synthesis of N-alkyl glycine amides as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem Lett18:3891-4 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Leukotriene A-4 hydrolase |
|---|
| Name: | Leukotriene A-4 hydrolase |
|---|
| Synonyms: | LKHA4_HUMAN | LTA-4 hydrolase | LTA4 | LTA4H | Leukotriene A(4) hydrolase | Leukotriene A-4 hydrolase (LTA4H) | Leukotriene A4 hydrolase |
|---|
| Type: | Hydrolase; metalloprotease |
|---|
| Mol. Mass.: | 69280.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant LTA4H. |
|---|
| Residue: | 611 |
|---|
| Sequence: | MPEIVDTCSLASPASVCRTKHLHLRCSVDFTRRTLTGTAALTVQSQEDNLRSLVLDTKDL
TIEKVVINGQEVKYALGERQSYKGSPMEISLPIALSKNQEIVIEISFETSPKSSALQWLT
PEQTSGKEHPYLFSQCQAIHCRAILPCQDTPSVKLTYTAEVSVPKELVALMSAIRDGETP
DPEDPSRKIYKFIQKVPIPCYLIALVVGALESRQIGPRTLVWSEKEQVEKSAYEFSETES
MLKIAEDLGGPYVWGQYDLLVLPPSFPYGGMENPCLTFVTPTLLAGDKSLSNVIAHEISH
SWTGNLVTNKTWDHFWLNEGHTVYLERHICGRLFGEKFRHFNALGGWGELQNSVKTFGET
HPFTKLVVDLTDIDPDVAYSSVPYEKGFALLFYLEQLLGGPEIFLGFLKAYVEKFSYKSI
TTDDWKDFLYSYFKDKVDVLNQVDWNAWLYSPGLPPIKPNYDMTLTNACIALSQRWITAK
EDDLNSFNATDLKDLSSHQLNEFLAQTLQRAPLPLGHIKRMQEVYNFNAINNSEIRFRWL
RLCIQSKWEDAIPLALKMATEQGRMKFTRPLFKDLAAFDKSHDQAVRTYQEHKASMHPVT
AMLVGKDLKVD
|
|
|
|---|
| BDBM50251385 |
|---|
| n/a |
|---|
| Name | BDBM50251385 |
|---|
| Synonyms: | 5-((methyl(3-(methyl(2-oxo-2-(4-phenoxyphenylamino)ethyl)amino)propyl)amino)methyl)furan-2-carboxylic acid | CHEMBL519094 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H29N3O5 |
|---|
| Mol. Mass. | 451.5149 |
|---|
| SMILES | CN(CCCN(C)Cc1ccc(o1)C(O)=O)CC(=O)Nc1ccc(Oc2ccccc2)cc1 |
|---|
| Structure | 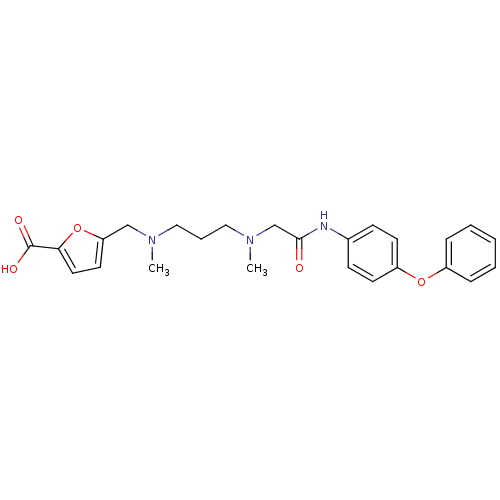
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ye, B; Bauman, J; Chen, M; Davey, D; Khim, SK; King, B; Kirkland, T; Kochanny, M; Liang, A; Lentz, D; May, K; Mendoza, L; Phillips, G; Selchau, V; Schlyer, S; Tseng, JL; Wei, RG; Ye, H; Parkinson, J; Guilford, WJ Synthesis of N-alkyl glycine amides as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem Lett18:3891-4 (2008) [PubMed] Article
Ye, B; Bauman, J; Chen, M; Davey, D; Khim, SK; King, B; Kirkland, T; Kochanny, M; Liang, A; Lentz, D; May, K; Mendoza, L; Phillips, G; Selchau, V; Schlyer, S; Tseng, JL; Wei, RG; Ye, H; Parkinson, J; Guilford, WJ Synthesis of N-alkyl glycine amides as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem Lett18:3891-4 (2008) [PubMed] Article