| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase Lck |
|---|
| Ligand | BDBM50264389 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_535209 (CHEMBL990805) |
|---|
| IC50 | 75±n/a nM |
|---|
| Citation |  Kunz, RK; Rumfelt, S; Chen, N; Zhang, D; Tasker, AS; Bürli, R; Hungate, R; Yu, V; Nguyen, Y; Whittington, DA; Meagher, KL; Plant, M; Tudor, Y; Schrag, M; Xu, Y; Ng, GY; Hu, E Discovery of amido-benzisoxazoles as potent c-Kit inhibitors. Bioorg Med Chem Lett18:5115-7 (2008) [PubMed] Article Kunz, RK; Rumfelt, S; Chen, N; Zhang, D; Tasker, AS; Bürli, R; Hungate, R; Yu, V; Nguyen, Y; Whittington, DA; Meagher, KL; Plant, M; Tudor, Y; Schrag, M; Xu, Y; Ng, GY; Hu, E Discovery of amido-benzisoxazoles as potent c-Kit inhibitors. Bioorg Med Chem Lett18:5115-7 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase Lck |
|---|
| Name: | Tyrosine-protein kinase Lck |
|---|
| Synonyms: | 2.7.10.2 | LCK | LCK_HUMAN | LSK | Leukocyte C-terminal Src kinase | Lymphocyte cell-specific protein-tyrosine kinase | Lymphocyte-specific protein tyrosine kinase | P56-LCK | Protein YT16 | Proto-oncogene Lck | Proto-oncogene tyrosine-protein kinase LCK | Src/Lck kinase | T cell-specific protein-tyrosine kinase |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 57987.83 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06239 |
|---|
| Residue: | 509 |
|---|
| Sequence: | MGCGCSSHPEDDWMENIDVCENCHYPIVPLDGKGTLLIRNGSEVRDPLVTYEGSNPPASP
LQDNLVIALHSYEPSHDGDLGFEKGEQLRILEQSGEWWKAQSLTTGQEGFIPFNFVAKAN
SLEPEPWFFKNLSRKDAERQLLAPGNTHGSFLIRESESTAGSFSLSVRDFDQNQGEVVKH
YKIRNLDNGGFYISPRITFPGLHELVRHYTNASDGLCTRLSRPCQTQKPQKPWWEDEWEV
PRETLKLVERLGAGQFGEVWMGYYNGHTKVAVKSLKQGSMSPDAFLAEANLMKQLQHQRL
VRLYAVVTQEPIYIITEYMENGSLVDFLKTPSGIKLTINKLLDMAAQIAEGMAFIEERNY
IHRDLRAANILVSDTLSCKIADFGLARLIEDNEYTAREGAKFPIKWTAPEAINYGTFTIK
SDVWSFGILLTEIVTHGRIPYPGMTNPEVIQNLERGYRMVRPDNCPEELYQLMRLCWKER
PEDRPTFDYLRSVLEDFFTATEGQYQPQP
|
|
|
|---|
| BDBM50264389 |
|---|
| n/a |
|---|
| Name | BDBM50264389 |
|---|
| Synonyms: | 6-chloro-N-(pyrimidin-5-yl)-3-(3-(trifluoromethyl)phenylamino)benzo[d]isoxazole-7-carboxamide | 6-chloro-N-pyrimidin-5-yl-3-{[3-(trifluoromethyl)phenyl]amino}-1,2-benzisoxazole-7-carboxamide | CHEMBL491429 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H11ClF3N5O2 |
|---|
| Mol. Mass. | 433.771 |
|---|
| SMILES | FC(F)(F)c1cccc(Nc2noc3c(C(=O)Nc4cncnc4)c(Cl)ccc23)c1 |
|---|
| Structure | 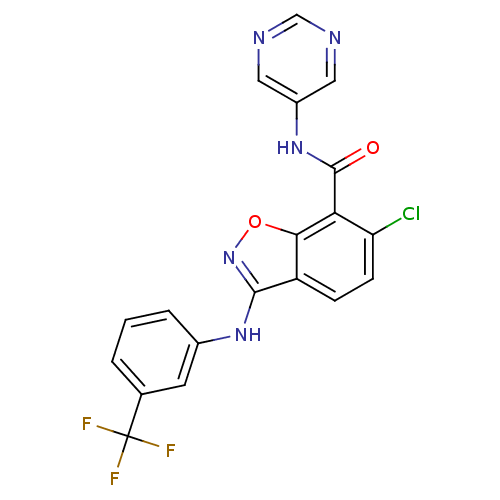
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kunz, RK; Rumfelt, S; Chen, N; Zhang, D; Tasker, AS; Bürli, R; Hungate, R; Yu, V; Nguyen, Y; Whittington, DA; Meagher, KL; Plant, M; Tudor, Y; Schrag, M; Xu, Y; Ng, GY; Hu, E Discovery of amido-benzisoxazoles as potent c-Kit inhibitors. Bioorg Med Chem Lett18:5115-7 (2008) [PubMed] Article
Kunz, RK; Rumfelt, S; Chen, N; Zhang, D; Tasker, AS; Bürli, R; Hungate, R; Yu, V; Nguyen, Y; Whittington, DA; Meagher, KL; Plant, M; Tudor, Y; Schrag, M; Xu, Y; Ng, GY; Hu, E Discovery of amido-benzisoxazoles as potent c-Kit inhibitors. Bioorg Med Chem Lett18:5115-7 (2008) [PubMed] Article