| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen phosphorylase, liver form |
|---|
| Ligand | BDBM50256669 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_515066 (CHEMBL1034808) |
|---|
| IC50 | 159±n/a nM |
|---|
| Citation |  Sparks, SM; Banker, P; Bickett, DM; Clancy, DC; Dickerson, SH; Garrido, DM; Golden, PL; Peat, AJ; Sheckler, LR; Tavares, FX; Thomson, SA; Weiel, JE Anthranilimide-based glycogen phosphorylase inhibitors for the treatment of Type 2 diabetes: 2. Optimization of serine and threonine ether amino acid residues. Bioorg Med Chem Lett19:981-5 (2009) [PubMed] Article Sparks, SM; Banker, P; Bickett, DM; Clancy, DC; Dickerson, SH; Garrido, DM; Golden, PL; Peat, AJ; Sheckler, LR; Tavares, FX; Thomson, SA; Weiel, JE Anthranilimide-based glycogen phosphorylase inhibitors for the treatment of Type 2 diabetes: 2. Optimization of serine and threonine ether amino acid residues. Bioorg Med Chem Lett19:981-5 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen phosphorylase, liver form |
|---|
| Name: | Glycogen phosphorylase, liver form |
|---|
| Synonyms: | Glycogen Phosphorylase (PYGL) | Glycogen Phosphorylase, liver form | Liver glycogen phosphorylase | PYGL | PYGL_HUMAN |
|---|
| Type: | Homodimer |
|---|
| Mol. Mass.: | 97153.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Dimers associate into a tetramer to form the enzymatically active phosphorylase A. |
|---|
| Residue: | 847 |
|---|
| Sequence: | MAKPLTDQEKRRQISIRGIVGVENVAELKKSFNRHLHFTLVKDRNVATTRDYYFALAHTV
RDHLVGRWIRTQQHYYDKCPKRVYYLSLEFYMGRTLQNTMINLGLQNACDEAIYQLGLDI
EELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEYGIFNQKIRDGWQVEEA
DDWLRYGNPWEKSRPEFMLPVHFYGKVEHTNTGTKWIDTQVVLALPYDTPVPGYMNNTVN
TMRLWSARAPNDFNLRDFNVGDYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFV
VAATLQDIIRRFKASKFGSTRGAGTVFDAFPDQVAIQLNDTHPALAIPELMRIFVDIEKL
PWSKAWELTQKTFAYTNHTVLPEALERWPVDLVEKLLPRHLEIIYEINQKHLDRIVALFP
KDVDRLRRMSLIEEEGSKRINMAHLCIVGSHAVNGVAKIHSDIVKTKVFKDFSELEPDKF
QNKTNGITPRRWLLLCNPGLAELIAEKIGEDYVKDLSQLTKLHSFLGDDVFLRELAKVKQ
ENKLKFSQFLETEYKVKINPSSMFDVQVKRIHEYKRQLLNCLHVITMYNRIKKDPKKLFV
PRTVIIGGKAAPGYHMAKMIIKLITSVADVVNNDPMVGSKLKVIFLENYRVSLAEKVIPA
TDLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENLFIFGMRIDDVA
ALDKKGYEAKEYYEALPELKLVIDQIDNGFFSPKQPDLFKDIINMLFYHDRFKVFADYEA
YVKCQDKVSQLYMNPKAWNTMVLKNIAASGKFSSDRTIKEYAQNIWNVEPSDLKISLSNE
SNKVNGN
|
|
|
|---|
| BDBM50256669 |
|---|
| n/a |
|---|
| Name | BDBM50256669 |
|---|
| Synonyms: | (2S,3R)-2-(3-(3-mesitylureido)-2-naphthamido)-3-(1-methylcyclopentyloxy)butanoic acid | CHEMBL474193 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H37N3O5 |
|---|
| Mol. Mass. | 531.6426 |
|---|
| SMILES | C[C@@H](OC1(C)CCCC1)[C@H](NC(=O)c1cc2ccccc2cc1NC(=O)Nc1c(C)cc(C)cc1C)C(O)=O |r| |
|---|
| Structure | 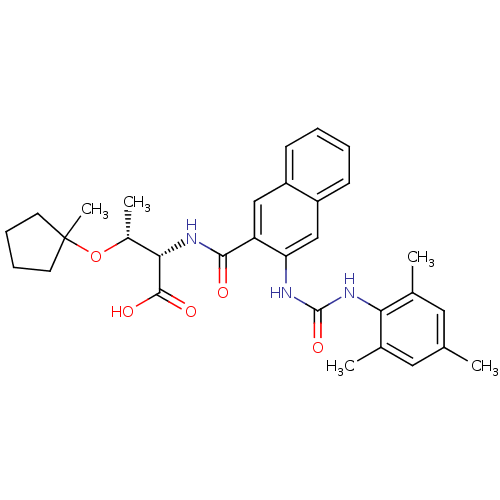
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sparks, SM; Banker, P; Bickett, DM; Clancy, DC; Dickerson, SH; Garrido, DM; Golden, PL; Peat, AJ; Sheckler, LR; Tavares, FX; Thomson, SA; Weiel, JE Anthranilimide-based glycogen phosphorylase inhibitors for the treatment of Type 2 diabetes: 2. Optimization of serine and threonine ether amino acid residues. Bioorg Med Chem Lett19:981-5 (2009) [PubMed] Article
Sparks, SM; Banker, P; Bickett, DM; Clancy, DC; Dickerson, SH; Garrido, DM; Golden, PL; Peat, AJ; Sheckler, LR; Tavares, FX; Thomson, SA; Weiel, JE Anthranilimide-based glycogen phosphorylase inhibitors for the treatment of Type 2 diabetes: 2. Optimization of serine and threonine ether amino acid residues. Bioorg Med Chem Lett19:981-5 (2009) [PubMed] Article