| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Aldo-keto reductase family 1 member C1 |
|---|
| Ligand | BDBM50219490 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_498201 (CHEMBL980733) |
|---|
| Ki | 4±n/a nM |
|---|
| Citation |  El-Kabbani, O; Scammells, PJ; Gosling, J; Dhagat, U; Endo, S; Matsunaga, T; Soda, M; Hara, A Structure-guided design, synthesis, and evaluation of salicylic acid-based inhibitors targeting a selectivity pocket in the active site of human 20alpha-hydroxysteroid dehydrogenase (AKR1C1). J Med Chem52:3259-64 (2009) [PubMed] Article El-Kabbani, O; Scammells, PJ; Gosling, J; Dhagat, U; Endo, S; Matsunaga, T; Soda, M; Hara, A Structure-guided design, synthesis, and evaluation of salicylic acid-based inhibitors targeting a selectivity pocket in the active site of human 20alpha-hydroxysteroid dehydrogenase (AKR1C1). J Med Chem52:3259-64 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Aldo-keto reductase family 1 member C1 |
|---|
| Name: | Aldo-keto reductase family 1 member C1 |
|---|
| Synonyms: | 20-alpha-HSD | 20-alpha-Hydroxysteroid Dehydrogenase (AKR1C1) | AK1C1_HUMAN | AKR1C1 | Aldo-keto reductase family 1 member C1 (AK1C1) | Aldo-keto reductase family 1 member C1 (AK1C1a) | Aldo-keto reductase family 1 member C1 (AKR1C1) | Chlordecone reductase homolog HAKRC | DDH | DDH1 | High-affinity hepatic bile acid-binding protein | Trans-1,2-dihydrobenzene-1,2-diol dehydrogenase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36793.97 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant AKR1C1 enzyme was expressed in E. coli. |
|---|
| Residue: | 323 |
|---|
| Sequence: | MDSKYQCVKLNDGHFMPVLGFGTYAPAEVPKSKALEATKLAIEAGFRHIDSAHLYNNEEQ
VGLAIRSKIADGSVKREDIFYTSKLWCNSHRPELVRPALERSLKNLQLDYVDLYLIHFPV
SVKPGEEVIPKDENGKILFDTVDLCATWEAVEKCKDAGLAKSIGVSNFNRRQLEMILNKP
GLKYKPVCNQVECHPYFNQRKLLDFCKSKDIVLVAYSALGSHREEPWVDPNSPVLLEDPV
LCALAKKHKRTPALIALRYQLQRGVVVLAKSYNEQRIRQNVQVFEFQLTSEEMKAIDGLN
RNVRYLTLDIFAGPPNYPFSDEY
|
|
|
|---|
| BDBM50219490 |
|---|
| n/a |
|---|
| Name | BDBM50219490 |
|---|
| Synonyms: | 3-Bromo-5-phenylsalicylc acid | 5-bromo-4-hydroxybiphenyl-3-carboxylic acid | CHEMBL387536 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H9BrO3 |
|---|
| Mol. Mass. | 293.113 |
|---|
| SMILES | OC(=O)c1cc(cc(Br)c1O)-c1ccccc1 |
|---|
| Structure | 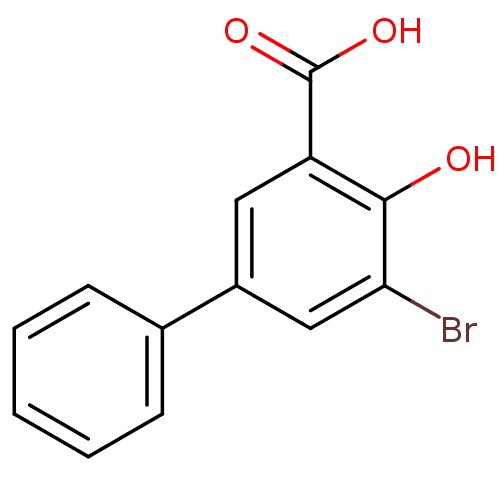
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 El-Kabbani, O; Scammells, PJ; Gosling, J; Dhagat, U; Endo, S; Matsunaga, T; Soda, M; Hara, A Structure-guided design, synthesis, and evaluation of salicylic acid-based inhibitors targeting a selectivity pocket in the active site of human 20alpha-hydroxysteroid dehydrogenase (AKR1C1). J Med Chem52:3259-64 (2009) [PubMed] Article
El-Kabbani, O; Scammells, PJ; Gosling, J; Dhagat, U; Endo, S; Matsunaga, T; Soda, M; Hara, A Structure-guided design, synthesis, and evaluation of salicylic acid-based inhibitors targeting a selectivity pocket in the active site of human 20alpha-hydroxysteroid dehydrogenase (AKR1C1). J Med Chem52:3259-64 (2009) [PubMed] Article