| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM35283 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_579855 (CHEMBL1063241) |
|---|
| IC50 | >200000±n/a nM |
|---|
| Citation |  Shi, Y; Zhang, J; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Sitkoff, D; Pudzianowski, AT; Chong, S; Klei, HE; Kish, K; Yanchunas, J; Liu, EC; Hartl, KS; Seiler, SM; Steinbacher, TE; Schumacher, WA; Atwal, KS; Stein, PD Cyanoguanidine-based lactam derivatives as a novel class of orally bioavailable factor Xa inhibitors. Bioorg Med Chem Lett19:4034-41 (2009) [PubMed] Article Shi, Y; Zhang, J; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Sitkoff, D; Pudzianowski, AT; Chong, S; Klei, HE; Kish, K; Yanchunas, J; Liu, EC; Hartl, KS; Seiler, SM; Steinbacher, TE; Schumacher, WA; Atwal, KS; Stein, PD Cyanoguanidine-based lactam derivatives as a novel class of orally bioavailable factor Xa inhibitors. Bioorg Med Chem Lett19:4034-41 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM35283 |
|---|
| n/a |
|---|
| Name | BDBM35283 |
|---|
| Synonyms: | CHEMBL551991 | cyanoguanidine, 3 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H28N6O3 |
|---|
| Mol. Mass. | 436.5068 |
|---|
| SMILES | Cc1cc2cc(ccc2o1)N=C(NC#N)N[C@H]1CCCCN(CC(=O)N2CCCC2)C1=O |r,w:10.11| |
|---|
| Structure | 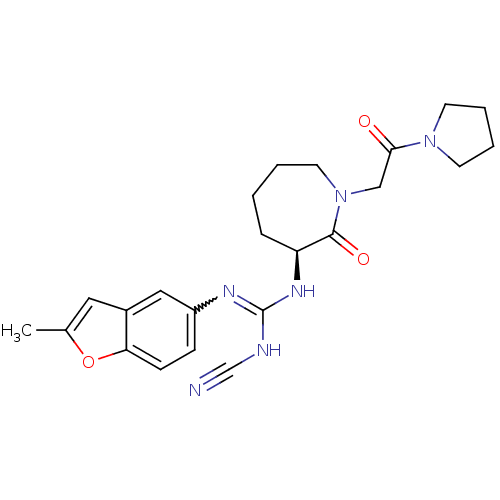
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shi, Y; Zhang, J; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Sitkoff, D; Pudzianowski, AT; Chong, S; Klei, HE; Kish, K; Yanchunas, J; Liu, EC; Hartl, KS; Seiler, SM; Steinbacher, TE; Schumacher, WA; Atwal, KS; Stein, PD Cyanoguanidine-based lactam derivatives as a novel class of orally bioavailable factor Xa inhibitors. Bioorg Med Chem Lett19:4034-41 (2009) [PubMed] Article
Shi, Y; Zhang, J; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Sitkoff, D; Pudzianowski, AT; Chong, S; Klei, HE; Kish, K; Yanchunas, J; Liu, EC; Hartl, KS; Seiler, SM; Steinbacher, TE; Schumacher, WA; Atwal, KS; Stein, PD Cyanoguanidine-based lactam derivatives as a novel class of orally bioavailable factor Xa inhibitors. Bioorg Med Chem Lett19:4034-41 (2009) [PubMed] Article