| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin E2 receptor EP4 subtype |
|---|
| Ligand | BDBM50308124 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_610063 (CHEMBL1072472) |
|---|
| Ki | 6.4±n/a nM |
|---|
| Citation |  Blouin, M; Han, Y; Burch, J; Farand, J; Mellon, C; Gaudreault, M; Wrona, M; Lévesque, JF; Denis, D; Mathieu, MC; Stocco, R; Vigneault, E; Therien, A; Clark, P; Rowland, S; Xu, D; O'Neill, G; Ducharme, Y; Friesen, R The discovery of 4-{1-[({2,5-dimethyl-4-[4-(trifluoromethyl)benzyl]-3-thienyl}carbonyl)amino]cyclopropyl}benzoic acid (MK-2894), a potent and selective prostaglandin E2 subtype 4 receptor antagonist. J Med Chem53:2227-38 (2010) [PubMed] Article Blouin, M; Han, Y; Burch, J; Farand, J; Mellon, C; Gaudreault, M; Wrona, M; Lévesque, JF; Denis, D; Mathieu, MC; Stocco, R; Vigneault, E; Therien, A; Clark, P; Rowland, S; Xu, D; O'Neill, G; Ducharme, Y; Friesen, R The discovery of 4-{1-[({2,5-dimethyl-4-[4-(trifluoromethyl)benzyl]-3-thienyl}carbonyl)amino]cyclopropyl}benzoic acid (MK-2894), a potent and selective prostaglandin E2 subtype 4 receptor antagonist. J Med Chem53:2227-38 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin E2 receptor EP4 subtype |
|---|
| Name: | Prostaglandin E2 receptor EP4 subtype |
|---|
| Synonyms: | PE2R4_HUMAN | PGE receptor EP4 subtype | PGE2 receptor EP4 subtype | PTGER2 | PTGER4 | Prostaglandin E2 receptor | Prostanoid EP4 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 53134.53 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35408 |
|---|
| Residue: | 488 |
|---|
| Sequence: | MSTPGVNSSASLSPDRLNSPVTIPAVMFIFGVVGNLVAIVVLCKSRKEQKETTFYTLVCG
LAVTDLLGTLLVSPVTIATYMKGQWPGGQPLCEYSTFILLFFSLSGLSIICAMSVERYLA
INHAYFYSHYVDKRLAGLTLFAVYASNVLFCALPNMGLGSSRLQYPDTWCFIDWTTNVTA
HAAYSYMYAGFSSFLILATVLCNVLVCGALLRMHRQFMRRTSLGTEQHHAAAAASVASRG
HPAASPALPRLSDFRRRRSFRRIAGAEIQMVILLIATSLVVLICSIPLVVRVFVNQLYQP
SLEREVSKNPDLQAIRIASVNPILDPWIYILLRKTVLSKAIEKIKCLFCRIGGSRRERSG
QHCSDSQRTSSAMSGHSRSFISRELKEISSTSQTLLPDLSLPDLSENGLGGRNLLPGVPG
MGLAQEDTTSLRTLRISETSDSSQGQDSESVLLVDEAGGSGRAGPAPKGSSLQVTFPSET
LNLSEKCI
|
|
|
|---|
| BDBM50308124 |
|---|
| n/a |
|---|
| Name | BDBM50308124 |
|---|
| Synonyms: | 4-[(1S)-1-({[4-(3-Chlorobenzyl)-3-thienyl]carbonyl}amino)ethyl]-benzoic Acid | CHEMBL599051 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H18ClNO3S |
|---|
| Mol. Mass. | 399.891 |
|---|
| SMILES | C[C@H](NC(=O)c1cscc1Cc1cccc(Cl)c1)c1ccc(cc1)C(O)=O |r| |
|---|
| Structure | 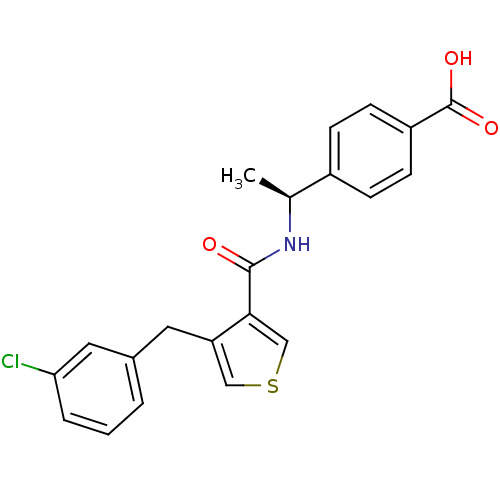
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Blouin, M; Han, Y; Burch, J; Farand, J; Mellon, C; Gaudreault, M; Wrona, M; Lévesque, JF; Denis, D; Mathieu, MC; Stocco, R; Vigneault, E; Therien, A; Clark, P; Rowland, S; Xu, D; O'Neill, G; Ducharme, Y; Friesen, R The discovery of 4-{1-[({2,5-dimethyl-4-[4-(trifluoromethyl)benzyl]-3-thienyl}carbonyl)amino]cyclopropyl}benzoic acid (MK-2894), a potent and selective prostaglandin E2 subtype 4 receptor antagonist. J Med Chem53:2227-38 (2010) [PubMed] Article
Blouin, M; Han, Y; Burch, J; Farand, J; Mellon, C; Gaudreault, M; Wrona, M; Lévesque, JF; Denis, D; Mathieu, MC; Stocco, R; Vigneault, E; Therien, A; Clark, P; Rowland, S; Xu, D; O'Neill, G; Ducharme, Y; Friesen, R The discovery of 4-{1-[({2,5-dimethyl-4-[4-(trifluoromethyl)benzyl]-3-thienyl}carbonyl)amino]cyclopropyl}benzoic acid (MK-2894), a potent and selective prostaglandin E2 subtype 4 receptor antagonist. J Med Chem53:2227-38 (2010) [PubMed] Article