| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin D2 receptor |
|---|
| Ligand | BDBM50319841 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_634906 (CHEMBL1120633) |
|---|
| Ki | 885±n/a nM |
|---|
| Citation |  Colucci, J; Boyd, M; Berthelette, C; Chiasson, JF; Wang, Z; Ducharme, Y; Friesen, R; Wrona, M; Levesque, JF; Denis, D; Mathieu, MC; Stocco, R; Therien, AG; Clarke, P; Rowland, S; Xu, D; Han, Y Discovery of 4-[1-[([1-[4-(trifluoromethyl)benzyl]-1H-indol-7-yl]carbonyl)amino]cyclopropyl]benzoic acid (MF-766), a highly potent and selective EP4 antagonist for treating inflammatory pain. Bioorg Med Chem Lett20:3760-3 (2010) [PubMed] Article Colucci, J; Boyd, M; Berthelette, C; Chiasson, JF; Wang, Z; Ducharme, Y; Friesen, R; Wrona, M; Levesque, JF; Denis, D; Mathieu, MC; Stocco, R; Therien, AG; Clarke, P; Rowland, S; Xu, D; Han, Y Discovery of 4-[1-[([1-[4-(trifluoromethyl)benzyl]-1H-indol-7-yl]carbonyl)amino]cyclopropyl]benzoic acid (MF-766), a highly potent and selective EP4 antagonist for treating inflammatory pain. Bioorg Med Chem Lett20:3760-3 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin D2 receptor |
|---|
| Name: | Prostaglandin D2 receptor |
|---|
| Synonyms: | PD2R_HUMAN | PTGDR | Prostaglandin D2 | Prostaglandin D2 receptor | Prostanoid DP receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40288.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q13258 |
|---|
| Residue: | 359 |
|---|
| Sequence: | MKSPFYRCQNTTSVEKGNSAVMGGVLFSTGLLGNLLALGLLARSGLGWCSRRPLRPLPSV
FYMLVCGLTVTDLLGKCLLSPVVLAAYAQNRSLRVLAPALDNSLCQAFAFFMSFFGLSST
LQLLAMALECWLSLGHPFFYRRHITLRLGALVAPVVSAFSLAFCALPFMGFGKFVQYCPG
TWCFIQMVHEEGSLSVLGYSVLYSSLMALLVLATVLCNLGAMRNLYAMHRRLQRHPRSCT
RDCAEPRADGREASPQPLEELDHLLLLALMTVLFTMCSLPVIYRAYYGAFKDVKEKNRTS
EEAEDLRALRFLSVISIVDPWIFIIFRSPVFRIFFHKIFIRPLRYRSRCSNSTNMESSL
|
|
|
|---|
| BDBM50319841 |
|---|
| n/a |
|---|
| Name | BDBM50319841 |
|---|
| Synonyms: | (S)-4-(1-(1-(3-(trifluoromethyl)benzyl)-1H-indole-7-carboxamido)ethyl)benzoic acid | CHEMBL1084551 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H21F3N2O3 |
|---|
| Mol. Mass. | 466.4517 |
|---|
| SMILES | C[C@H](NC(=O)c1cccc2ccn(Cc3cccc(c3)C(F)(F)F)c12)c1ccc(cc1)C(O)=O |r| |
|---|
| Structure | 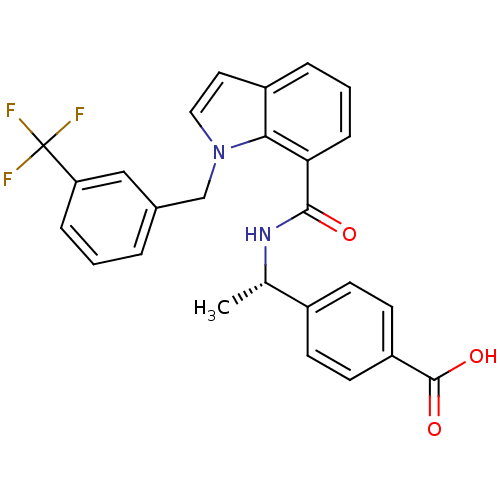
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Colucci, J; Boyd, M; Berthelette, C; Chiasson, JF; Wang, Z; Ducharme, Y; Friesen, R; Wrona, M; Levesque, JF; Denis, D; Mathieu, MC; Stocco, R; Therien, AG; Clarke, P; Rowland, S; Xu, D; Han, Y Discovery of 4-[1-[([1-[4-(trifluoromethyl)benzyl]-1H-indol-7-yl]carbonyl)amino]cyclopropyl]benzoic acid (MF-766), a highly potent and selective EP4 antagonist for treating inflammatory pain. Bioorg Med Chem Lett20:3760-3 (2010) [PubMed] Article
Colucci, J; Boyd, M; Berthelette, C; Chiasson, JF; Wang, Z; Ducharme, Y; Friesen, R; Wrona, M; Levesque, JF; Denis, D; Mathieu, MC; Stocco, R; Therien, AG; Clarke, P; Rowland, S; Xu, D; Han, Y Discovery of 4-[1-[([1-[4-(trifluoromethyl)benzyl]-1H-indol-7-yl]carbonyl)amino]cyclopropyl]benzoic acid (MF-766), a highly potent and selective EP4 antagonist for treating inflammatory pain. Bioorg Med Chem Lett20:3760-3 (2010) [PubMed] Article