| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50320180 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_635412 (CHEMBL1120212) |
|---|
| IC50 | 3.7±n/a nM |
|---|
| Citation |  Madsen-Duggan, CB; Debenham, JS; Walsh, TF; Yan, L; Huo, P; Wang, J; Tong, X; Lao, J; Fong, TM; Xiao, JC; Huang, CR; Shen, CP; Stribling, DS; Shearman, LP; Strack, AM; Goulet, MT; Hale, JJ Dihydro-pyrano[2,3-b]pyridines and tetrahydro-1,8-naphthyridines as CB1 receptor inverse agonists: synthesis, SAR and biological evaluation. Bioorg Med Chem Lett20:3750-4 (2010) [PubMed] Article Madsen-Duggan, CB; Debenham, JS; Walsh, TF; Yan, L; Huo, P; Wang, J; Tong, X; Lao, J; Fong, TM; Xiao, JC; Huang, CR; Shen, CP; Stribling, DS; Shearman, LP; Strack, AM; Goulet, MT; Hale, JJ Dihydro-pyrano[2,3-b]pyridines and tetrahydro-1,8-naphthyridines as CB1 receptor inverse agonists: synthesis, SAR and biological evaluation. Bioorg Med Chem Lett20:3750-4 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50320180 |
|---|
| n/a |
|---|
| Name | BDBM50320180 |
|---|
| Synonyms: | 6-(4-CHLOROPHENYL)-7-(2,4-DICHLOROPHENYL)-N-(HYDROXYMETHYL)-1,2,2-TRIMETHYL-1,2,3,4-TETRAHYDRO-1,8-NAPHTHYRIDINE-4-CARBOXAMIDE (ENANTIOMERIC MIX) | 6-(4-chlorophenyl)-7-(2,4-dichlorophenyl)-N-(hydroxymethyl)-1,2,2-trimethyl-1,2,3,4-tetrahydro-1,8-naphthyridine-4-carboxamide | CHEMBL1082330 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H24Cl3N3O2 |
|---|
| Mol. Mass. | 504.836 |
|---|
| SMILES | CN1c2nc(-c3ccc(Cl)cc3Cl)c(cc2C(CC1(C)C)C(=O)NCO)-c1ccc(Cl)cc1 |
|---|
| Structure | 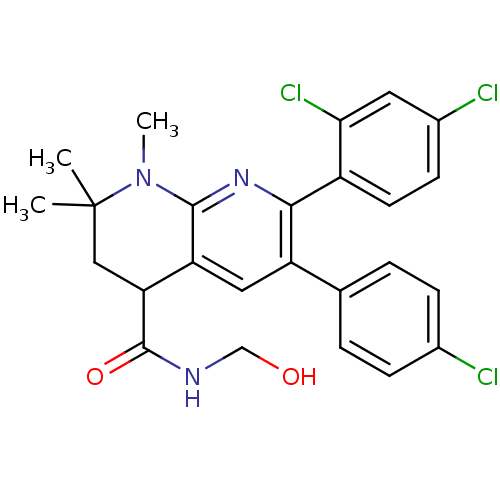
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Madsen-Duggan, CB; Debenham, JS; Walsh, TF; Yan, L; Huo, P; Wang, J; Tong, X; Lao, J; Fong, TM; Xiao, JC; Huang, CR; Shen, CP; Stribling, DS; Shearman, LP; Strack, AM; Goulet, MT; Hale, JJ Dihydro-pyrano[2,3-b]pyridines and tetrahydro-1,8-naphthyridines as CB1 receptor inverse agonists: synthesis, SAR and biological evaluation. Bioorg Med Chem Lett20:3750-4 (2010) [PubMed] Article
Madsen-Duggan, CB; Debenham, JS; Walsh, TF; Yan, L; Huo, P; Wang, J; Tong, X; Lao, J; Fong, TM; Xiao, JC; Huang, CR; Shen, CP; Stribling, DS; Shearman, LP; Strack, AM; Goulet, MT; Hale, JJ Dihydro-pyrano[2,3-b]pyridines and tetrahydro-1,8-naphthyridines as CB1 receptor inverse agonists: synthesis, SAR and biological evaluation. Bioorg Med Chem Lett20:3750-4 (2010) [PubMed] Article