| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polyphenol oxidase 2 |
|---|
| Ligand | BDBM50324035 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_647410 (CHEMBL1217571) |
|---|
| Ki | 9850±n/a nM |
|---|
| Citation |  Bandgar, BP; Totre, JV; Gawande, SS; Khobragade, CN; Warangkar, SC; Kadam, PD Synthesis of novel 3,5-diaryl pyrazole derivatives using combinatorial chemistry as inhibitors of tyrosinase as well as potent anticancer, anti-inflammatory agents. Bioorg Med Chem18:6149-55 (2010) [PubMed] Article Bandgar, BP; Totre, JV; Gawande, SS; Khobragade, CN; Warangkar, SC; Kadam, PD Synthesis of novel 3,5-diaryl pyrazole derivatives using combinatorial chemistry as inhibitors of tyrosinase as well as potent anticancer, anti-inflammatory agents. Bioorg Med Chem18:6149-55 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polyphenol oxidase 2 |
|---|
| Name: | Polyphenol oxidase 2 |
|---|
| Synonyms: | Monophenol monooxygenase | Monophenol oxidase | PPO2 | PPO2_AGABI | Tyrosinase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 63923.66 |
|---|
| Organism: | Agaricus bisporus (Common mushroom) |
|---|
| Description: | O42713 |
|---|
| Residue: | 556 |
|---|
| Sequence: | MSLIATVGPTGGVKNRLNIVDFVKNEKFFTLYVRSLELLQAKEQHDYSSFFQLAGIHGLP
FTEWAKERPSMNLYKAGYCTHGQVLFPTWHRTYLSVLEQILQGAAIEVAKKFTSNQTDWV
QAAQDLRQPYWDWGFELMPPDEVIKNEEVNITNYDGKKISVKNPILRYHFHPIDPSFKPY
GDFATWRTTVRNPDRNRREDIPGLIKKMRLEEGQIREKTYNMLKFNDAWERFSNHGISDD
QHANSLESVHDDIHVMVGYGKIEGHMDHPFFAAFDPIFWLHHTNVDRLLSLWKAINPDVW
VTSGRNRDGTMGIAPNAQINSETPLEPFYQSGDKVWTSASLADTARLGYSYPDFDKLVGG
TKELIRDAIDDLIDERYGSKPSSGARNTAFDLLADFKGITKEHKEDLKMYDWTIHVAFKK
FELKESFSLLFYFASDGGDYDQENCFVGSINAFRGTAPETCANCQDNENLIQEGFIHLNH
YLARDLESFEPQDVHKFLKEKGLSYKLYSRGDKPLTSLSVKIEGRPLHLPPGEHRPKYDH
TQARVVFDDVAVHVIN
|
|
|
|---|
| BDBM50324035 |
|---|
| n/a |
|---|
| Name | BDBM50324035 |
|---|
| Synonyms: | 2-[5-(2-Chloro-4-nitro-phenyl)-1H-pyrazol-3-yl]-6-(2-hydroxymethyl-1-methyl-pyrrolidin-3-yl)-3,5-dimethoxyphenol | CHEMBL1214239 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25ClN4O6 |
|---|
| Mol. Mass. | 488.921 |
|---|
| SMILES | COc1cc(OC)c(-c2cc(n[nH]2)-c2ccc(cc2Cl)[N+]([O-])=O)c(O)c1C1CCN(C)C1CO |
|---|
| Structure | 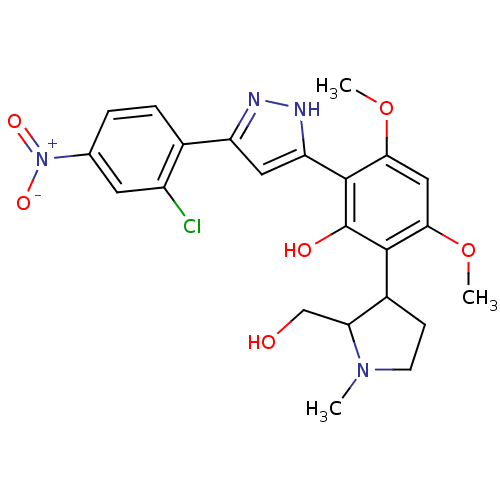
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bandgar, BP; Totre, JV; Gawande, SS; Khobragade, CN; Warangkar, SC; Kadam, PD Synthesis of novel 3,5-diaryl pyrazole derivatives using combinatorial chemistry as inhibitors of tyrosinase as well as potent anticancer, anti-inflammatory agents. Bioorg Med Chem18:6149-55 (2010) [PubMed] Article
Bandgar, BP; Totre, JV; Gawande, SS; Khobragade, CN; Warangkar, SC; Kadam, PD Synthesis of novel 3,5-diaryl pyrazole derivatives using combinatorial chemistry as inhibitors of tyrosinase as well as potent anticancer, anti-inflammatory agents. Bioorg Med Chem18:6149-55 (2010) [PubMed] Article