| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Multidrug resistance-associated protein 1 |
|---|
| Ligand | BDBM50338974 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_727306 (CHEMBL1687517) |
|---|
| IC50 | 50000±n/a nM |
|---|
| Citation |  Pick, A; Müller, H; Mayer, R; Haenisch, B; Pajeva, IK; Weigt, M; Bönisch, H; Müller, CE; Wiese, M Structure-activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorg Med Chem19:2090-102 (2011) [PubMed] Article Pick, A; Müller, H; Mayer, R; Haenisch, B; Pajeva, IK; Weigt, M; Bönisch, H; Müller, CE; Wiese, M Structure-activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorg Med Chem19:2090-102 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Multidrug resistance-associated protein 1 |
|---|
| Name: | Multidrug resistance-associated protein 1 |
|---|
| Synonyms: | 7.6.2.2 | 7.6.2.3 | ABCC1 | ATP-binding cassette sub-family C member 1 | ATP-binding cassette, sub-family C, member 1 isoform 1 | Glutathione-S-conjugate-translocating ATPase ABCC1 | LTC4 transporter | Leukotriene C(4) transporter | MRP | MRP1 | MRP1_HUMAN |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 171597.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33527 |
|---|
| Residue: | 1531 |
|---|
| Sequence: | MALRGFCSADGSDPLWDWNVTWNTSNPDFTKCFQNTVLVWVPCFYLWACFPFYFLYLSRH
DRGYIQMTPLNKTKTALGFLLWIVCWADLFYSFWERSRGIFLAPVFLVSPTLLGITMLLA
TFLIQLERRKGVQSSGIMLTFWLVALVCALAILRSKIMTALKEDAQVDLFRDITFYVYFS
LLLIQLVLSCFSDRSPLFSETIHDPNPCPESSASFLSRITFWWITGLIVRGYRQPLEGSD
LWSLNKEDTSEQVVPVLVKNWKKECAKTRKQPVKVVYSSKDPAQPKESSKVDANEEVEAL
IVKSPQKEWNPSLFKVLYKTFGPYFLMSFFFKAIHDLMMFSGPQILKLLIKFVNDTKAPD
WQGYFYTVLLFVTACLQTLVLHQYFHICFVSGMRIKTAVIGAVYRKALVITNSARKSSTV
GEIVNLMSVDAQRFMDLATYINMIWSAPLQVILALYLLWLNLGPSVLAGVAVMVLMVPVN
AVMAMKTKTYQVAHMKSKDNRIKLMNEILNGIKVLKLYAWELAFKDKVLAIRQEELKVLK
KSAYLSAVGTFTWVCTPFLVALCTFAVYVTIDENNILDAQTAFVSLALFNILRFPLNILP
MVISSIVQASVSLKRLRIFLSHEELEPDSIERRPVKDGGGTNSITVRNATFTWARSDPPT
LNGITFSIPEGALVAVVGQVGCGKSSLLSALLAEMDKVEGHVAIKGSVAYVPQQAWIQND
SLRENILFGCQLEEPYYRSVIQACALLPDLEILPSGDRTEIGEKGVNLSGGQKQRVSLAR
AVYSNADIYLFDDPLSAVDAHVGKHIFENVIGPKGMLKNKTRILVTHSMSYLPQVDVIIV
MSGGKISEMGSYQELLARDGAFAEFLRTYASTEQEQDAEENGVTGVSGPGKEAKQMENGM
LVTDSAGKQLQRQLSSSSSYSGDISRHHNSTAELQKAEAKKEETWKLMEADKAQTGQVKL
SVYWDYMKAIGLFISFLSIFLFMCNHVSALASNYWLSLWTDDPIVNGTQEHTKVRLSVYG
ALGISQGIAVFGYSMAVSIGGILASRCLHVDLLHSILRSPMSFFERTPSGNLVNRFSKEL
DTVDSMIPEVIKMFMGSLFNVIGACIVILLATPIAAIIIPPLGLIYFFVQRFYVASSRQL
KRLESVSRSPVYSHFNETLLGVSVIRAFEEQERFIHQSDLKVDENQKAYYPSIVANRWLA
VRLECVGNCIVLFAALFAVISRHSLSAGLVGLSVSYSLQVTTYLNWLVRMSSEMETNIVA
VERLKEYSETEKEAPWQIQETAPPSSWPQVGRVEFRNYCLRYREDLDFVLRHINVTINGG
EKVGIVGRTGAGKSSLTLGLFRINESAEGEIIIDGINIAKIGLHDLRFKITIIPQDPVLF
SGSLRMNLDPFSQYSDEEVWTSLELAHLKDFVSALPDKLDHECAEGGENLSVGQRQLVCL
ARALLRKTKILVLDEATAAVDLETDDLIQSTIRTQFEDCTVLTIAHRLNTIMDYTRVIVL
DKGEIQEYGAPSDLLQQRGLFYSMAKDAGLV
|
|
|
|---|
| BDBM50338974 |
|---|
| n/a |
|---|
| Name | BDBM50338974 |
|---|
| Synonyms: | 2-(3,4-Dimethoxy-phenyl)-5-hydroxy-3,7-dimethoxy-chromen-4-one | 2-(3,4-dimethoxyphenyl)-5-hydroxy-3,7-dimethoxy-4H-chromen-4-one | 5-hydroxy-3,7,3,4-tetramethoxyflavone | CHEMBL77966 | NSC-61837 | quercetin tetramethyl ether | quercitin-3,7,3',4'-tetramethyl ether | retusin |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H18O7 |
|---|
| Mol. Mass. | 358.342 |
|---|
| SMILES | COc1cc(O)c2c(c1)oc(-c1ccc(OC)c(OC)c1)c(OC)c2=O |
|---|
| Structure | 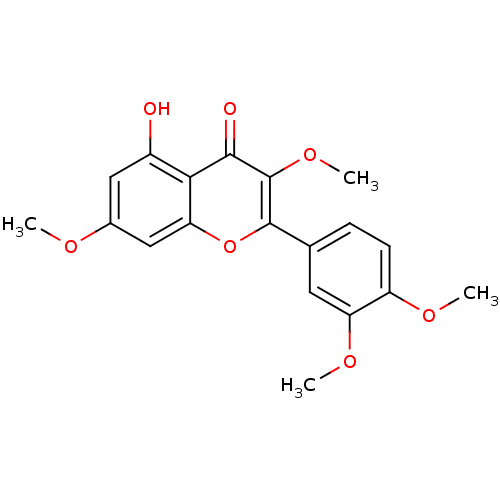
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pick, A; Müller, H; Mayer, R; Haenisch, B; Pajeva, IK; Weigt, M; Bönisch, H; Müller, CE; Wiese, M Structure-activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorg Med Chem19:2090-102 (2011) [PubMed] Article
Pick, A; Müller, H; Mayer, R; Haenisch, B; Pajeva, IK; Weigt, M; Bönisch, H; Müller, CE; Wiese, M Structure-activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorg Med Chem19:2090-102 (2011) [PubMed] Article