| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50347939 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_755921 (CHEMBL1803730) |
|---|
| IC50 | >8900±n/a nM |
|---|
| Citation |  Wu, T; Nagle, A; Kuhen, K; Gagaring, K; Borboa, R; Francek, C; Chen, Z; Plouffe, D; Goh, A; Lakshminarayana, SB; Wu, J; Ang, HQ; Zeng, P; Kang, ML; Tan, W; Tan, M; Ye, N; Lin, X; Caldwell, C; Ek, J; Skolnik, S; Liu, F; Wang, J; Chang, J; Li, C; Hollenbeck, T; Tuntland, T; Isbell, J; Fischli, C; Brun, R; Rottmann, M; Dartois, V; Keller, T; Diagana, T; Winzeler, E; Glynne, R; Tully, DC; Chatterjee, AK Imidazolopiperazines: hit to lead optimization of new antimalarial agents. J Med Chem54:5116-30 (2011) [PubMed] Article Wu, T; Nagle, A; Kuhen, K; Gagaring, K; Borboa, R; Francek, C; Chen, Z; Plouffe, D; Goh, A; Lakshminarayana, SB; Wu, J; Ang, HQ; Zeng, P; Kang, ML; Tan, W; Tan, M; Ye, N; Lin, X; Caldwell, C; Ek, J; Skolnik, S; Liu, F; Wang, J; Chang, J; Li, C; Hollenbeck, T; Tuntland, T; Isbell, J; Fischli, C; Brun, R; Rottmann, M; Dartois, V; Keller, T; Diagana, T; Winzeler, E; Glynne, R; Tully, DC; Chatterjee, AK Imidazolopiperazines: hit to lead optimization of new antimalarial agents. J Med Chem54:5116-30 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50347939 |
|---|
| n/a |
|---|
| Name | BDBM50347939 |
|---|
| Synonyms: | CHEMBL1800137 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H23F2N5O |
|---|
| Mol. Mass. | 411.4477 |
|---|
| SMILES | CC(C)(N)C(=O)N1CCn2c(C1)nc(c2Nc1ccc(F)cc1)-c1ccc(F)cc1 |
|---|
| Structure | 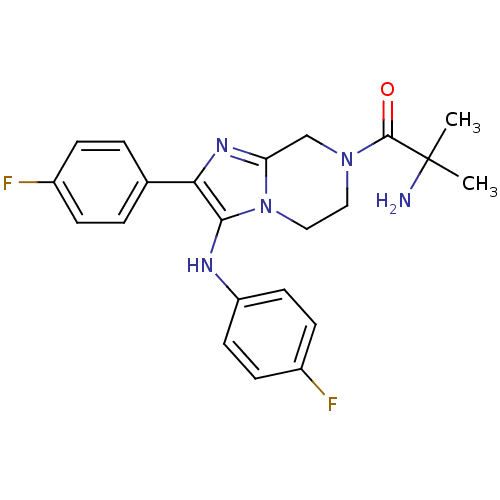
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wu, T; Nagle, A; Kuhen, K; Gagaring, K; Borboa, R; Francek, C; Chen, Z; Plouffe, D; Goh, A; Lakshminarayana, SB; Wu, J; Ang, HQ; Zeng, P; Kang, ML; Tan, W; Tan, M; Ye, N; Lin, X; Caldwell, C; Ek, J; Skolnik, S; Liu, F; Wang, J; Chang, J; Li, C; Hollenbeck, T; Tuntland, T; Isbell, J; Fischli, C; Brun, R; Rottmann, M; Dartois, V; Keller, T; Diagana, T; Winzeler, E; Glynne, R; Tully, DC; Chatterjee, AK Imidazolopiperazines: hit to lead optimization of new antimalarial agents. J Med Chem54:5116-30 (2011) [PubMed] Article
Wu, T; Nagle, A; Kuhen, K; Gagaring, K; Borboa, R; Francek, C; Chen, Z; Plouffe, D; Goh, A; Lakshminarayana, SB; Wu, J; Ang, HQ; Zeng, P; Kang, ML; Tan, W; Tan, M; Ye, N; Lin, X; Caldwell, C; Ek, J; Skolnik, S; Liu, F; Wang, J; Chang, J; Li, C; Hollenbeck, T; Tuntland, T; Isbell, J; Fischli, C; Brun, R; Rottmann, M; Dartois, V; Keller, T; Diagana, T; Winzeler, E; Glynne, R; Tully, DC; Chatterjee, AK Imidazolopiperazines: hit to lead optimization of new antimalarial agents. J Med Chem54:5116-30 (2011) [PubMed] Article