| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50354894 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_772750 (CHEMBL1837362) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Johnson, PS; Ryckmans, T; Bryans, J; Beal, DM; Dack, KN; Feeder, N; Harrison, A; Lewis, M; Mason, HJ; Mills, J; Newman, J; Pasquinet, C; Rawson, DJ; Roberts, LR; Russell, R; Spark, D; Stobie, A; Underwood, TJ; Ward, R; Wheeler, S Discovery of PF-184563, a potent and selective V1a antagonist for the treatment of dysmenorrhoea. The influence of compound flexibility on microsomal stability. Bioorg Med Chem Lett21:5684-7 (2011) [PubMed] Article Johnson, PS; Ryckmans, T; Bryans, J; Beal, DM; Dack, KN; Feeder, N; Harrison, A; Lewis, M; Mason, HJ; Mills, J; Newman, J; Pasquinet, C; Rawson, DJ; Roberts, LR; Russell, R; Spark, D; Stobie, A; Underwood, TJ; Ward, R; Wheeler, S Discovery of PF-184563, a potent and selective V1a antagonist for the treatment of dysmenorrhoea. The influence of compound flexibility on microsomal stability. Bioorg Med Chem Lett21:5684-7 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50354894 |
|---|
| n/a |
|---|
| Name | BDBM50354894 |
|---|
| Synonyms: | CHEMBL1837037 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H23ClN6 |
|---|
| Mol. Mass. | 394.901 |
|---|
| SMILES | CN1Cc2nnc(C3CCN(CC3)c3ccccn3)n2-c2ccc(Cl)cc2C1 |
|---|
| Structure | 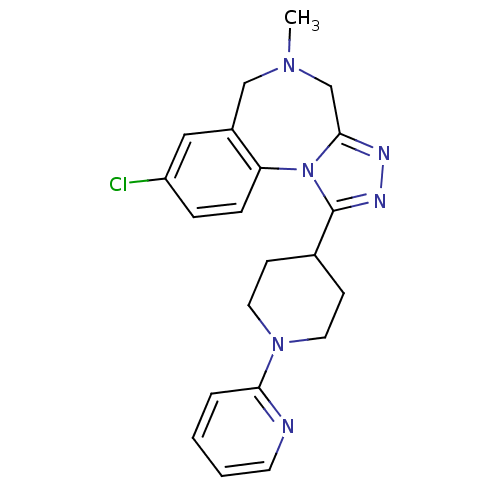
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Johnson, PS; Ryckmans, T; Bryans, J; Beal, DM; Dack, KN; Feeder, N; Harrison, A; Lewis, M; Mason, HJ; Mills, J; Newman, J; Pasquinet, C; Rawson, DJ; Roberts, LR; Russell, R; Spark, D; Stobie, A; Underwood, TJ; Ward, R; Wheeler, S Discovery of PF-184563, a potent and selective V1a antagonist for the treatment of dysmenorrhoea. The influence of compound flexibility on microsomal stability. Bioorg Med Chem Lett21:5684-7 (2011) [PubMed] Article
Johnson, PS; Ryckmans, T; Bryans, J; Beal, DM; Dack, KN; Feeder, N; Harrison, A; Lewis, M; Mason, HJ; Mills, J; Newman, J; Pasquinet, C; Rawson, DJ; Roberts, LR; Russell, R; Spark, D; Stobie, A; Underwood, TJ; Ward, R; Wheeler, S Discovery of PF-184563, a potent and selective V1a antagonist for the treatment of dysmenorrhoea. The influence of compound flexibility on microsomal stability. Bioorg Med Chem Lett21:5684-7 (2011) [PubMed] Article