

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Substance-K receptor | ||
| Ligand | BDBM50045037 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_141787 (CHEMBL745325) | ||
| IC50 | 65±n/a nM | ||
| Citation |  Kucharczyk, N; Thurieau, C; Paladino, J; Morris, AD; Bonnet, J; Canet, E; Krause, JE; Regoli, D; Couture, R; Fauchère, JL Tetrapeptide tachykinin antagonists: synthesis and modulation of the physicochemical and pharmacological properties of a new series of partially cyclic analogs. J Med Chem36:1654-61 (1993) [PubMed] Kucharczyk, N; Thurieau, C; Paladino, J; Morris, AD; Bonnet, J; Canet, E; Krause, JE; Regoli, D; Couture, R; Fauchère, JL Tetrapeptide tachykinin antagonists: synthesis and modulation of the physicochemical and pharmacological properties of a new series of partially cyclic analogs. J Med Chem36:1654-61 (1993) [PubMed] | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Substance-K receptor | |||
| Name: | Substance-K receptor | ||
| Synonyms: | NK-2 receptor | NK-2R | NK2R_RAT | Neurokinin 2 receptor | Neurokinin A receptor | Neurokinin NK2 | SKR | Substance-K receptor | Tac2r | Tachykinin receptor 2 | Tacr2 | ||
| Type: | Enzyme Catalytic Domain | ||
| Mol. Mass.: | 43868.63 | ||
| Organism: | Rattus norvegicus (Rat) | ||
| Description: | Neurokinin NK2 TACR2 Murine::P16610 | ||
| Residue: | 390 | ||
| Sequence: |
| ||
| BDBM50045037 | |||
| n/a | |||
| Name | BDBM50045037 | ||
| Synonyms: | CHEMBL3144352 | N-Benzyl-2-[2-[2-(3,6-dioxo-2,5-diaza-tricyclo[6.2.2.0*2,7*]dodec-4-yl)-acetylamino]-3-(1H-indol-3-yl)-propionylamino]-N-methyl-3-phenyl-propionamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C40H44N6O5 | ||
| Mol. Mass. | 688.8146 | ||
| SMILES | CN(Cc1ccccc1)C(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)NC(=O)C[C@@H]1NC(=O)[C@@H]2C3CCC(CC3)N2C1=O |r,wU:41.44,37.40,wD:11.11,22.24,THB:39:41:44.43:46.47,(10.7,-11.55,;10.4,-10.04,;11.55,-9.02,;13.01,-9.52,;14.17,-8.5,;15.63,-8.99,;15.93,-10.5,;14.77,-11.52,;13.31,-11.03,;8.94,-9.55,;8.63,-8.04,;7.78,-10.56,;8.08,-12.07,;6.93,-13.09,;7.23,-14.6,;6.07,-15.62,;4.61,-15.12,;4.31,-13.61,;5.47,-12.6,;6.32,-10.07,;5.16,-11.09,;4.52,-12.48,;3.71,-10.59,;3.4,-9.08,;4.56,-8.07,;6.06,-8.4,;6.85,-7.08,;5.83,-5.92,;6.01,-4.39,;4.78,-3.47,;3.36,-4.08,;3.18,-5.61,;4.42,-6.53,;2.55,-11.61,;1.09,-11.12,;.79,-9.61,;-.07,-12.13,;-1.53,-11.64,;-1.83,-10.13,;-3.29,-9.63,;-3.59,-8.12,;-4.44,-10.65,;-5.8,-10.12,;-7.3,-10.77,;-7.1,-12.18,;-5.54,-11.51,;-5.28,-9.58,;-5.73,-8.44,;-4.14,-12.16,;-2.68,-12.66,;-2.38,-14.17,)| | ||
| Structure | 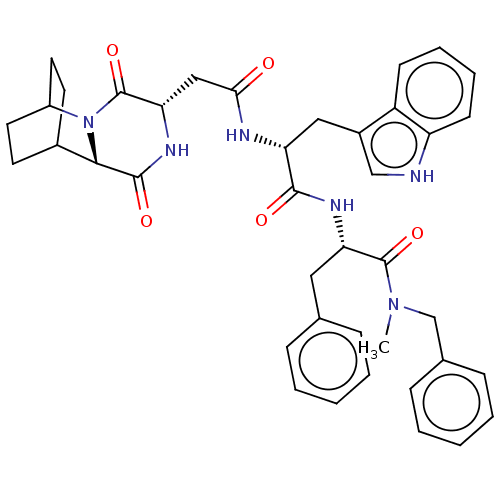
| ||