| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM18971 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48828 (CHEMBL661801) |
|---|
| Ki | 11±n/a nM |
|---|
| Citation |  Pruitt, JR; Pinto, DJ; Galemmo, RA; Alexander, RS; Rossi, KA; Wells, BL; Drummond, S; Bostrom, LL; Burdick, D; Bruckner, R; Chen, H; Smallwood, A; Wong, PC; Wright, MR; Bai, S; Luettgen, JM; Knabb, RM; Lam, PY; Wexler, RR Discovery of 1-(2-aminomethylphenyl)-3-trifluoromethyl-N- [3-fluoro-2'-(aminosulfonyl)[1,1'-biphenyl)]-4-yl]-1H-pyrazole-5-carboxyamide (DPC602), a potent, selective, and orally bioavailable factor Xa inhibitor(1). J Med Chem46:5298-315 (2003) [PubMed] Article Pruitt, JR; Pinto, DJ; Galemmo, RA; Alexander, RS; Rossi, KA; Wells, BL; Drummond, S; Bostrom, LL; Burdick, D; Bruckner, R; Chen, H; Smallwood, A; Wong, PC; Wright, MR; Bai, S; Luettgen, JM; Knabb, RM; Lam, PY; Wexler, RR Discovery of 1-(2-aminomethylphenyl)-3-trifluoromethyl-N- [3-fluoro-2'-(aminosulfonyl)[1,1'-biphenyl)]-4-yl]-1H-pyrazole-5-carboxyamide (DPC602), a potent, selective, and orally bioavailable factor Xa inhibitor(1). J Med Chem46:5298-315 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM18971 |
|---|
| n/a |
|---|
| Name | BDBM18971 |
|---|
| Synonyms: | 1-(4-methoxyphenyl)-3-methyl-N-[4-(2-sulfamoylphenyl)phenyl]-1H-pyrazole-5-carboxamide | CHEMBL139788 | pyrazole-based inhibitor, 22a |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H22N4O4S |
|---|
| Mol. Mass. | 462.521 |
|---|
| SMILES | COc1ccc(cc1)-n1nc(C)cc1C(=O)Nc1ccc(cc1)-c1ccccc1S(N)(=O)=O |
|---|
| Structure | 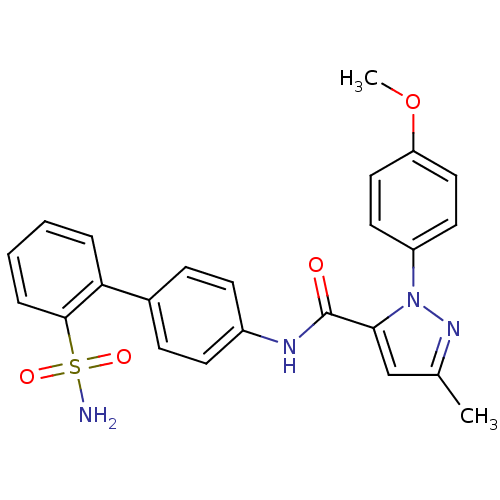
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pruitt, JR; Pinto, DJ; Galemmo, RA; Alexander, RS; Rossi, KA; Wells, BL; Drummond, S; Bostrom, LL; Burdick, D; Bruckner, R; Chen, H; Smallwood, A; Wong, PC; Wright, MR; Bai, S; Luettgen, JM; Knabb, RM; Lam, PY; Wexler, RR Discovery of 1-(2-aminomethylphenyl)-3-trifluoromethyl-N- [3-fluoro-2'-(aminosulfonyl)[1,1'-biphenyl)]-4-yl]-1H-pyrazole-5-carboxyamide (DPC602), a potent, selective, and orally bioavailable factor Xa inhibitor(1). J Med Chem46:5298-315 (2003) [PubMed] Article
Pruitt, JR; Pinto, DJ; Galemmo, RA; Alexander, RS; Rossi, KA; Wells, BL; Drummond, S; Bostrom, LL; Burdick, D; Bruckner, R; Chen, H; Smallwood, A; Wong, PC; Wright, MR; Bai, S; Luettgen, JM; Knabb, RM; Lam, PY; Wexler, RR Discovery of 1-(2-aminomethylphenyl)-3-trifluoromethyl-N- [3-fluoro-2'-(aminosulfonyl)[1,1'-biphenyl)]-4-yl]-1H-pyrazole-5-carboxyamide (DPC602), a potent, selective, and orally bioavailable factor Xa inhibitor(1). J Med Chem46:5298-315 (2003) [PubMed] Article