| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasminogen |
|---|
| Ligand | BDBM50216557 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_447831 (CHEMBL898081) |
|---|
| Ki | >22000±n/a nM |
|---|
| Citation |  Qiao, JX; Chang, CH; Cheney, DL; Morin, PE; Wang, GZ; King, SR; Wang, TC; Rendina, AR; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY SAR and X-ray structures of enantiopure 1,2-cis-(1R,2S)-cyclopentyldiamine and cyclohexyldiamine derivatives as inhibitors of coagulation Factor Xa. Bioorg Med Chem Lett17:4419-27 (2007) [PubMed] Article Qiao, JX; Chang, CH; Cheney, DL; Morin, PE; Wang, GZ; King, SR; Wang, TC; Rendina, AR; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY SAR and X-ray structures of enantiopure 1,2-cis-(1R,2S)-cyclopentyldiamine and cyclohexyldiamine derivatives as inhibitors of coagulation Factor Xa. Bioorg Med Chem Lett17:4419-27 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Plasminogen |
|---|
| Name: | Plasminogen |
|---|
| Synonyms: | Activation peptide | Angiostatin | PLG | PLMN_HUMAN | Plasmin | Plasmin heavy chain A | Plasmin heavy chain A, short form | Plasmin light chain B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 90579.18 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 810 |
|---|
| Sequence: | MEHKEVVLLLLLFLKSGQGEPLDDYVNTQGASLFSVTKKQLGAGSIEECAAKCEEDEEFT
CRAFQYHSKEQQCVIMAENRKSSIIIRMRDVVLFEKKVYLSECKTGNGKNYRGTMSKTKN
GITCQKWSSTSPHRPRFSPATHPSEGLEENYCRNPDNDPQGPWCYTTDPEKRYDYCDILE
CEEECMHCSGENYDGKISKTMSGLECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRE
LRPWCFTTDPNKRWELCDIPRCTTPPPSSGPTYQCLKGTGENYRGNVAVTVSGHTCQHWS
AQTPHTHNRTPENFPCKNLDENYCRNPDGKRAPWCHTTNSQVRWEYCKIPSCDSSPVSTE
QLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRHQKTPENYPNAG
LTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKCSGTEASVVAPPPVVLLPDVETPSEED
CMFGNGKGYRGKRATTVTGTPCQDWAAQEPHRHSIFTPETNPRAGLEKNYCRNPDGDVGG
PWCYTTNPRKLYDYCDVPQCAAPSFDCGKPQVEPKKCPGRVVGGCVAHPHSWPWQVSLRT
RFGMHFCGGTLISPEWVLTAAHCLEKSPRPSSYKVILGAHQEVNLEPHVQEIEVSRLFLE
PTRKDIALLKLSSPAVITDKVIPACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQ
LPVIENKVCNRYEFLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSW
GLGCARPNKPGVYVRVSRFVTWIEGVMRNN
|
|
|
|---|
| BDBM50216557 |
|---|
| n/a |
|---|
| Name | BDBM50216557 |
|---|
| Synonyms: | 5-chloro-N-((1R,2S)-2-(4-(2-oxopiperidin-1-yl)benzamido)cyclopentyl)thiophene-2-carboxamide | CHEMBL399593 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H24ClN3O3S |
|---|
| Mol. Mass. | 445.962 |
|---|
| SMILES | Clc1ccc(s1)C(=O)N[C@@H]1CCC[C@@H]1NC(=O)c1ccc(cc1)N1CCCCC1=O |
|---|
| Structure | 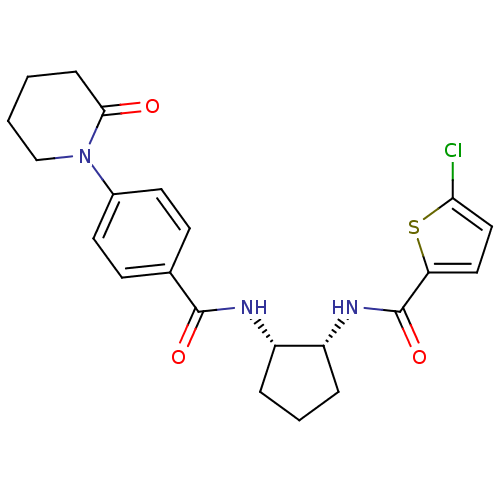
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Qiao, JX; Chang, CH; Cheney, DL; Morin, PE; Wang, GZ; King, SR; Wang, TC; Rendina, AR; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY SAR and X-ray structures of enantiopure 1,2-cis-(1R,2S)-cyclopentyldiamine and cyclohexyldiamine derivatives as inhibitors of coagulation Factor Xa. Bioorg Med Chem Lett17:4419-27 (2007) [PubMed] Article
Qiao, JX; Chang, CH; Cheney, DL; Morin, PE; Wang, GZ; King, SR; Wang, TC; Rendina, AR; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY SAR and X-ray structures of enantiopure 1,2-cis-(1R,2S)-cyclopentyldiamine and cyclohexyldiamine derivatives as inhibitors of coagulation Factor Xa. Bioorg Med Chem Lett17:4419-27 (2007) [PubMed] Article