| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM21221 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_450736 (CHEMBL901019) |
|---|
| Ki | 401±n/a nM |
|---|
| Citation |  Da Settimo, F; Primofiore, G; Taliani, S; Marini, AM; La Motta, C; Simorini, F; Salerno, S; Sergianni, V; Tuccinardi, T; Martinelli, A; Cosimelli, B; Greco, G; Novellino, E; Ciampi, O; Trincavelli, ML; Martini, C 5-amino-2-phenyl[1,2,3]triazolo[1,2-a][1,2,4]benzotriazin-1-one: a versatile scaffold to obtain potent and selective A3 adenosine receptor antagonists. J Med Chem50:5676-84 (2007) [PubMed] Article Da Settimo, F; Primofiore, G; Taliani, S; Marini, AM; La Motta, C; Simorini, F; Salerno, S; Sergianni, V; Tuccinardi, T; Martinelli, A; Cosimelli, B; Greco, G; Novellino, E; Ciampi, O; Trincavelli, ML; Martini, C 5-amino-2-phenyl[1,2,3]triazolo[1,2-a][1,2,4]benzotriazin-1-one: a versatile scaffold to obtain potent and selective A3 adenosine receptor antagonists. J Med Chem50:5676-84 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM21221 |
|---|
| n/a |
|---|
| Name | BDBM21221 |
|---|
| Synonyms: | (2S,3S,4R,5R)-5-(2-chloro-6-{[(3-iodophenyl)methyl]amino}-9H-purin-9-yl)-3,4-dihydroxy-N-methyloxolane-2-carboxamide | 2-Cl-IB-MECA | 2-chloro-N6-(3-iodobenzyl)adenosine-5-N-methylcarboxamide | C-IBzA-MU | C1-IB-MECA | CHEMBL431733 | CI-IB-MECA | Cl-IB-MECA | US20230364057, Compound 76 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H18ClIN6O4 |
|---|
| Mol. Mass. | 544.731 |
|---|
| SMILES | CNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 |
|---|
| Structure | 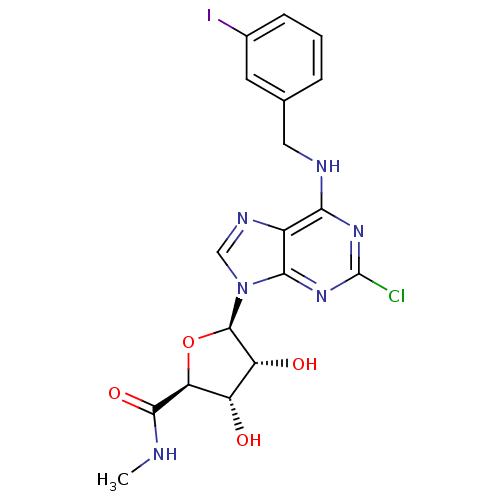
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Da Settimo, F; Primofiore, G; Taliani, S; Marini, AM; La Motta, C; Simorini, F; Salerno, S; Sergianni, V; Tuccinardi, T; Martinelli, A; Cosimelli, B; Greco, G; Novellino, E; Ciampi, O; Trincavelli, ML; Martini, C 5-amino-2-phenyl[1,2,3]triazolo[1,2-a][1,2,4]benzotriazin-1-one: a versatile scaffold to obtain potent and selective A3 adenosine receptor antagonists. J Med Chem50:5676-84 (2007) [PubMed] Article
Da Settimo, F; Primofiore, G; Taliani, S; Marini, AM; La Motta, C; Simorini, F; Salerno, S; Sergianni, V; Tuccinardi, T; Martinelli, A; Cosimelli, B; Greco, G; Novellino, E; Ciampi, O; Trincavelli, ML; Martini, C 5-amino-2-phenyl[1,2,3]triazolo[1,2-a][1,2,4]benzotriazin-1-one: a versatile scaffold to obtain potent and selective A3 adenosine receptor antagonists. J Med Chem50:5676-84 (2007) [PubMed] Article