| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50382412 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_815430 (CHEMBL2027082) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Barawkar, DA; Meru, A; Bandyopadhyay, A; Banerjee, A; Deshpande, AM; Athare, C; Koduru, C; Khose, G; Gundu, J; Mahajan, K; Patil, P; Kandalkar, SR; Niranjan, S; Bhosale, S; De, S; Mukhopadhyay, S; Chaudhary, S; Koul, S; Singh, U; Chugh, A; Palle, VP; Mookhtiar, KA; Vacca, J; Chakravarty, PK; Nargund, RP; Wright, SD; Roy, S; Graziano, MP; Singh, SB; Cully, D; Cai, TQ Potent and Selective Inhibitors of Long Chain l-2-Hydroxy Acid Oxidase Reduced Blood Pressure in DOCA Salt-Treated Rats. ACS Med Chem Lett2:919-923 (2011) [PubMed] Article Barawkar, DA; Meru, A; Bandyopadhyay, A; Banerjee, A; Deshpande, AM; Athare, C; Koduru, C; Khose, G; Gundu, J; Mahajan, K; Patil, P; Kandalkar, SR; Niranjan, S; Bhosale, S; De, S; Mukhopadhyay, S; Chaudhary, S; Koul, S; Singh, U; Chugh, A; Palle, VP; Mookhtiar, KA; Vacca, J; Chakravarty, PK; Nargund, RP; Wright, SD; Roy, S; Graziano, MP; Singh, SB; Cully, D; Cai, TQ Potent and Selective Inhibitors of Long Chain l-2-Hydroxy Acid Oxidase Reduced Blood Pressure in DOCA Salt-Treated Rats. ACS Med Chem Lett2:919-923 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50382412 |
|---|
| n/a |
|---|
| Name | BDBM50382412 |
|---|
| Synonyms: | CHEMBL2022570 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H15FN2O2 |
|---|
| Mol. Mass. | 310.3223 |
|---|
| SMILES | Cc1[nH]nc(C(O)=O)c1Cc1cccc(c1)-c1ccc(F)cc1 |
|---|
| Structure | 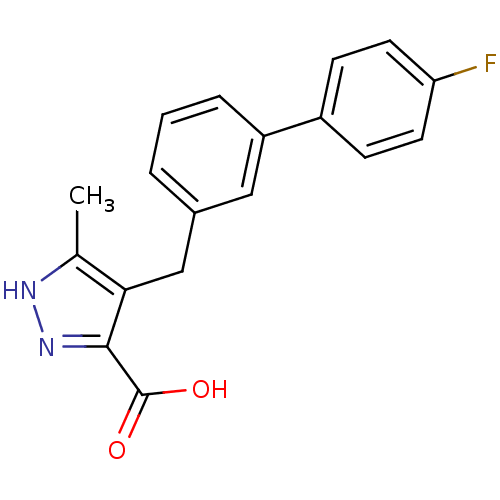
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Barawkar, DA; Meru, A; Bandyopadhyay, A; Banerjee, A; Deshpande, AM; Athare, C; Koduru, C; Khose, G; Gundu, J; Mahajan, K; Patil, P; Kandalkar, SR; Niranjan, S; Bhosale, S; De, S; Mukhopadhyay, S; Chaudhary, S; Koul, S; Singh, U; Chugh, A; Palle, VP; Mookhtiar, KA; Vacca, J; Chakravarty, PK; Nargund, RP; Wright, SD; Roy, S; Graziano, MP; Singh, SB; Cully, D; Cai, TQ Potent and Selective Inhibitors of Long Chain l-2-Hydroxy Acid Oxidase Reduced Blood Pressure in DOCA Salt-Treated Rats. ACS Med Chem Lett2:919-923 (2011) [PubMed] Article
Barawkar, DA; Meru, A; Bandyopadhyay, A; Banerjee, A; Deshpande, AM; Athare, C; Koduru, C; Khose, G; Gundu, J; Mahajan, K; Patil, P; Kandalkar, SR; Niranjan, S; Bhosale, S; De, S; Mukhopadhyay, S; Chaudhary, S; Koul, S; Singh, U; Chugh, A; Palle, VP; Mookhtiar, KA; Vacca, J; Chakravarty, PK; Nargund, RP; Wright, SD; Roy, S; Graziano, MP; Singh, SB; Cully, D; Cai, TQ Potent and Selective Inhibitors of Long Chain l-2-Hydroxy Acid Oxidase Reduced Blood Pressure in DOCA Salt-Treated Rats. ACS Med Chem Lett2:919-923 (2011) [PubMed] Article