| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 11-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Ligand | BDBM50359991 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_821681 (CHEMBL2038877) |
|---|
| IC50 | 0.4±n/a nM |
|---|
| Citation |  Mpetga, JD; Shen, Y; Tane, P; Li, SF; He, HP; Wabo, HK; Tene, M; Leng, Y; Hao, XJ Cycloartane and friedelane triterpenoids from the leaves of Caloncoba glauca and their evaluation for inhibition of 11ß-hydroxysteroid dehydrogenases. J Nat Prod75:599-604 (2012) [PubMed] Article Mpetga, JD; Shen, Y; Tane, P; Li, SF; He, HP; Wabo, HK; Tene, M; Leng, Y; Hao, XJ Cycloartane and friedelane triterpenoids from the leaves of Caloncoba glauca and their evaluation for inhibition of 11ß-hydroxysteroid dehydrogenases. J Nat Prod75:599-604 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 11-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Name: | 11-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Synonyms: | 11-DH2 | 11-beta-HSD2 | 11-beta-Hydroxysteroid Dehydrogenase 2 (11-beta-HSD2) | 11-beta-hydroxysteroid dehydrogenase | 11-beta-hydroxysteroid dehydrogenase 2 | 11-beta-hydroxysteroid dehydrogenase type 2 | 11-beta-hydroxysteroid dehydrogenase type 2 (11-beta-HSD2) | Corticosteroid 11-beta-dehydrogenase isozyme 2 | DHI2_HUMAN | HSD11B2 | HSD11K | NAD-dependent 11-beta-hydroxysteroid dehydrogenase | SDR9C3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44141.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Purified recombinant human 11beta-HSD2. |
|---|
| Residue: | 405 |
|---|
| Sequence: | MERWPWPSGGAWLLVAARALLQLLRSDLRLGRPLLAALALLAALDWLCQRLLPPPAALAV
LAAAGWIALSRLARPQRLPVATRAVLITGCDSGFGKETAKKLDSMGFTVLATVLELNSPG
AIELRTCCSPRLRLLQMDLTKPGDISRVLEFTKAHTTSTGLWGLVNNAGHNEVVADAELS
PVATFRSCMEVNFFGALELTKGLLPLLRSSRGRIVTVGSPAGDMPYPCLGAYGTSKAAVA
LLMDTFSCELLPWGVKVSIIQPGCFKTESVRNVGQWEKRKQLLLANLPQELLQAYGKDYI
EHLHGQFLHSLRLAMSDLTPVVDAITDALLAARPRRRYYPGQGLGLMYFIHYYLPEGLRR
RFLQAFFISHCLPRALQPGQPGTTPPQDAAQDPNLSPGPSPAVAR
|
|
|
|---|
| BDBM50359991 |
|---|
| n/a |
|---|
| Name | BDBM50359991 |
|---|
| Synonyms: | CHEMBL1927953 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C42H62O16 |
|---|
| Mol. Mass. | 822.9321 |
|---|
| SMILES | CC1(C)[C@H](CC[C@@]2(C)[C@H]1CC[C@]1(C)[C@@H]2C(=O)C=C2[C@@H]3C[C@](C)(CC[C@]3(C)CC[C@@]12C)C(O)=O)O[C@@H]1O[C@@H]([C@@H](O)[C@H](O[C@@H]2O[C@@H]([C@@H](O)[C@H](O)[C@H]2O)C(O)=O)[C@H]1O)C(O)=O |r,t:18| |
|---|
| Structure | 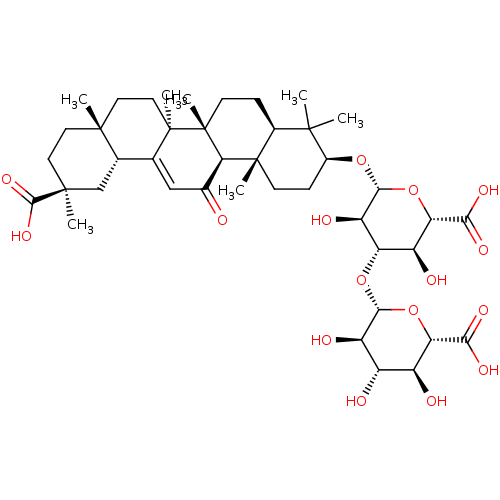
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mpetga, JD; Shen, Y; Tane, P; Li, SF; He, HP; Wabo, HK; Tene, M; Leng, Y; Hao, XJ Cycloartane and friedelane triterpenoids from the leaves of Caloncoba glauca and their evaluation for inhibition of 11ß-hydroxysteroid dehydrogenases. J Nat Prod75:599-604 (2012) [PubMed] Article
Mpetga, JD; Shen, Y; Tane, P; Li, SF; He, HP; Wabo, HK; Tene, M; Leng, Y; Hao, XJ Cycloartane and friedelane triterpenoids from the leaves of Caloncoba glauca and their evaluation for inhibition of 11ß-hydroxysteroid dehydrogenases. J Nat Prod75:599-604 (2012) [PubMed] Article