| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM85166 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2538 (CHEMBL616888) |
|---|
| Ki | 50.12±n/a nM |
|---|
| Citation |  Gaster, LM; Blaney, FE; Davies, S; Duckworth, DM; Ham, P; Jenkins, S; Jennings, AJ; Joiner, GF; King, FD; Mulholland, KR; Wyman, PA; Hagan, JJ; Hatcher, J; Jones, BJ; Middlemiss, DN; Price, GW; Riley, G; Roberts, C; Routledge, C; Selkirk, J; Slade, PD The selective 5-HT1B receptor inverse agonist 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro- spiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289) potently blocks terminal 5-HT autoreceptor function both in vitro and in vivo. J Med Chem41:1218-35 (1998) [PubMed] Article Gaster, LM; Blaney, FE; Davies, S; Duckworth, DM; Ham, P; Jenkins, S; Jennings, AJ; Joiner, GF; King, FD; Mulholland, KR; Wyman, PA; Hagan, JJ; Hatcher, J; Jones, BJ; Middlemiss, DN; Price, GW; Riley, G; Roberts, C; Routledge, C; Selkirk, J; Slade, PD The selective 5-HT1B receptor inverse agonist 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro- spiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289) potently blocks terminal 5-HT autoreceptor function both in vitro and in vivo. J Med Chem41:1218-35 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM85166 |
|---|
| n/a |
|---|
| Name | BDBM85166 |
|---|
| Synonyms: | CAS_3292447 | NSC_3292447 | SB 216641 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H30N4O4 |
|---|
| Mol. Mass. | 486.5622 |
|---|
| SMILES | COc1ccc(NC(=O)c2ccc(cc2)-c2ccc(cc2C)-c2noc(C)n2)cc1OCCN(C)C |
|---|
| Structure | 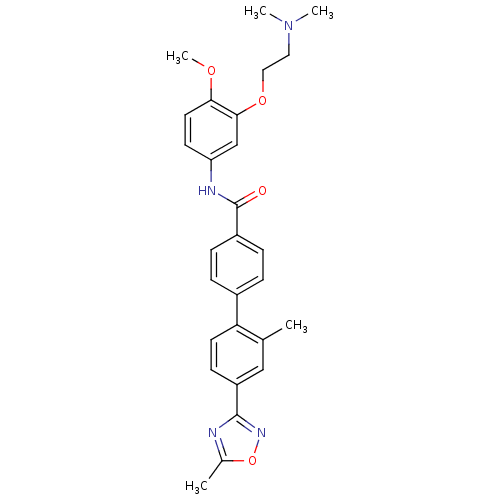
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gaster, LM; Blaney, FE; Davies, S; Duckworth, DM; Ham, P; Jenkins, S; Jennings, AJ; Joiner, GF; King, FD; Mulholland, KR; Wyman, PA; Hagan, JJ; Hatcher, J; Jones, BJ; Middlemiss, DN; Price, GW; Riley, G; Roberts, C; Routledge, C; Selkirk, J; Slade, PD The selective 5-HT1B receptor inverse agonist 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro- spiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289) potently blocks terminal 5-HT autoreceptor function both in vitro and in vivo. J Med Chem41:1218-35 (1998) [PubMed] Article
Gaster, LM; Blaney, FE; Davies, S; Duckworth, DM; Ham, P; Jenkins, S; Jennings, AJ; Joiner, GF; King, FD; Mulholland, KR; Wyman, PA; Hagan, JJ; Hatcher, J; Jones, BJ; Middlemiss, DN; Price, GW; Riley, G; Roberts, C; Routledge, C; Selkirk, J; Slade, PD The selective 5-HT1B receptor inverse agonist 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro- spiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289) potently blocks terminal 5-HT autoreceptor function both in vitro and in vivo. J Med Chem41:1218-35 (1998) [PubMed] Article