

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Microtubule-associated serine/threonine-protein kinase 3 | ||
| Ligand | BDBM50439425 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_980178 (CHEMBL2424216) | ||
| IC50 | 740±n/a nM | ||
| Citation |  Li, B; Cociorva, OM; Nomanbhoy, T; Weissig, H; Li, Q; Nakamura, K; Liyanage, M; Zhang, MC; Shih, AY; Aban, A; Hu, Y; Cajica, J; Pham, L; Kozarich, JW; Shreder, KR Hit-to-lead optimization and kinase selectivity of imidazo[1,2-a]quinoxalin-4-amine derived JNK1 inhibitors. Bioorg Med Chem Lett23:5217-22 (2013) [PubMed] Article Li, B; Cociorva, OM; Nomanbhoy, T; Weissig, H; Li, Q; Nakamura, K; Liyanage, M; Zhang, MC; Shih, AY; Aban, A; Hu, Y; Cajica, J; Pham, L; Kozarich, JW; Shreder, KR Hit-to-lead optimization and kinase selectivity of imidazo[1,2-a]quinoxalin-4-amine derived JNK1 inhibitors. Bioorg Med Chem Lett23:5217-22 (2013) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Microtubule-associated serine/threonine-protein kinase 3 | |||
| Name: | Microtubule-associated serine/threonine-protein kinase 3 | ||
| Synonyms: | KIAA0561 | MAST3 | MAST3_HUMAN | ||
| Type: | PROTEIN | ||
| Mol. Mass.: | 143157.77 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | ChEMBL_107645 | ||
| Residue: | 1309 | ||
| Sequence: |
| ||
| BDBM50439425 | |||
| n/a | |||
| Name | BDBM50439425 | ||
| Synonyms: | CHEMBL2420555 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C26H26FN5O2 | ||
| Mol. Mass. | 459.5153 | ||
| SMILES | O[C@H]1CC[C@@H](CC1)Nc1nc2ccc(cc2n2ccnc12)C(=O)NC1(CC1)c1ccc(F)cc1 |r,wU:4.7,wD:1.0,(5.97,-27.29,;7.31,-28.06,;7.31,-29.6,;8.64,-30.37,;9.97,-29.6,;9.97,-28.06,;8.64,-27.29,;11.31,-30.37,;12.64,-29.6,;13.97,-30.37,;15.31,-29.6,;16.64,-30.37,;17.98,-29.6,;17.98,-28.06,;16.64,-27.29,;15.31,-28.06,;13.97,-27.29,;13.65,-25.78,;12.12,-25.62,;11.5,-27.03,;12.64,-28.06,;19.31,-27.29,;19.31,-25.75,;20.64,-28.06,;21.98,-27.29,;22.97,-26.11,;21.45,-25.84,;23.24,-28.17,;23.1,-29.7,;24.37,-30.59,;25.76,-29.94,;27.02,-30.82,;25.89,-28.4,;24.63,-27.52,)| | ||
| Structure | 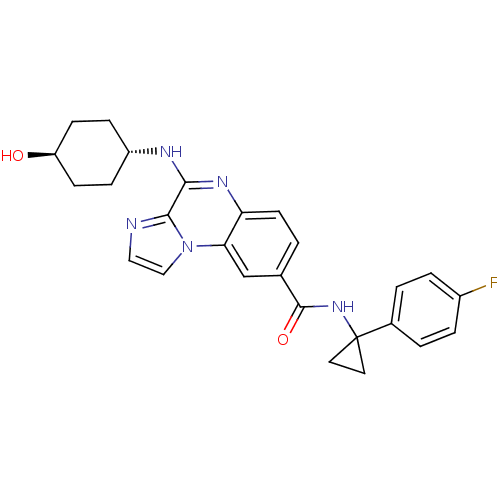
| ||