

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Tyrosine-protein kinase ITK/TSK | ||
| Ligand | BDBM50037062 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1435180 (CHEMBL3389531) | ||
| Ki | 177±n/a nM | ||
| Citation |  Trani, G; Barker, JJ; Bromidge, SM; Brookfield, FA; Burch, JD; Chen, Y; Eigenbrot, C; Heifetz, A; Ismaili, MH; Johnson, A; Kr�lle, TM; MacKinnon, CH; Maghames, R; McEwan, PA; Montalbetti, CA; Ortwine, DF; P�rez-Fuertes, Y; Vaidya, DG; Wang, X; Zarrin, AA; Pei, Z Design, synthesis and structure-activity relationships of a novel class of sulfonylpyridine inhibitors of Interleukin-2 inducible T-cell kinase (ITK). Bioorg Med Chem Lett24:5818-23 (2014) [PubMed] Article Trani, G; Barker, JJ; Bromidge, SM; Brookfield, FA; Burch, JD; Chen, Y; Eigenbrot, C; Heifetz, A; Ismaili, MH; Johnson, A; Kr�lle, TM; MacKinnon, CH; Maghames, R; McEwan, PA; Montalbetti, CA; Ortwine, DF; P�rez-Fuertes, Y; Vaidya, DG; Wang, X; Zarrin, AA; Pei, Z Design, synthesis and structure-activity relationships of a novel class of sulfonylpyridine inhibitors of Interleukin-2 inducible T-cell kinase (ITK). Bioorg Med Chem Lett24:5818-23 (2014) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Tyrosine-protein kinase ITK/TSK | |||
| Name: | Tyrosine-protein kinase ITK/TSK | ||
| Synonyms: | EMT | ITK | ITK_HUMAN | Kinase EMT | LYK | T-cell-specific kinase | Tyrosine-protein kinase ITK | Tyrosine-protein kinase ITK (ITK) | Tyrosine-protein kinase Lyk | ||
| Type: | Protein | ||
| Mol. Mass.: | 71839.20 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q08881 | ||
| Residue: | 620 | ||
| Sequence: |
| ||
| BDBM50037062 | |||
| n/a | |||
| Name | BDBM50037062 | ||
| Synonyms: | CHEMBL3355312 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C20H24N6O3S | ||
| Mol. Mass. | 428.508 | ||
| SMILES | Cc1cc(Nc2cc(nc(N[C@H]3CC[C@H](O)CC3)n2)S(=O)(=O)c2ccccc2)n[nH]1 |r,wU:11.10,wD:14.14,(15.1,-16.36,;13.56,-16.21,;12.54,-17.36,;11.12,-16.73,;9.79,-17.5,;9.8,-19.04,;8.47,-19.81,;8.47,-21.35,;9.8,-22.12,;11.14,-21.35,;12.47,-22.12,;12.47,-23.66,;13.81,-24.42,;13.8,-25.96,;12.47,-26.73,;12.46,-28.27,;11.13,-25.96,;11.14,-24.42,;11.13,-19.8,;7.12,-22.12,;7.89,-23.45,;6.35,-23.44,;5.8,-21.35,;5.81,-19.81,;4.48,-19.04,;3.14,-19.81,;3.14,-21.36,;4.48,-22.12,;11.28,-15.2,;12.79,-14.88,)| | ||
| Structure | 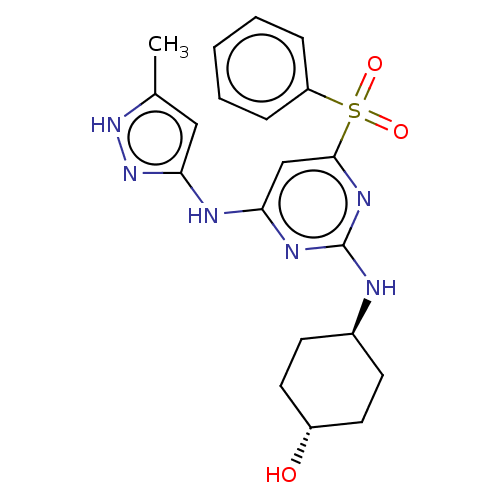
| ||