| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein kinase C alpha type |
|---|
| Ligand | BDBM50045370 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1459005 (CHEMBL3369598) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Xiao, D; Zhu, X; Sofolarides, M; Degrado, S; Shao, N; Rao, A; Chen, X; Aslanian, R; Fossetta, J; Tian, F; Trivedi, P; Lundell, D; Palani, A Discovery of a novel series of potent MK2 non-ATP competitive inhibitors using 1,2-substituted azoles as cis-amide isosteres. Bioorg Med Chem Lett24:3609-13 (2014) [PubMed] Article Xiao, D; Zhu, X; Sofolarides, M; Degrado, S; Shao, N; Rao, A; Chen, X; Aslanian, R; Fossetta, J; Tian, F; Trivedi, P; Lundell, D; Palani, A Discovery of a novel series of potent MK2 non-ATP competitive inhibitors using 1,2-substituted azoles as cis-amide isosteres. Bioorg Med Chem Lett24:3609-13 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein kinase C alpha type |
|---|
| Name: | Protein kinase C alpha type |
|---|
| Synonyms: | KPCA_HUMAN | PKC alpha | PKC alpha and beta-2 | PKC-A | PKC-alpha | PKCA | PRKACA | PRKCA | Protein Kinase C- alpha | Protein kinase C alpha | Protein kinase C alpha (PKCa) | Protein kinase C alpha type (PKC-A) | Protein kinase C alpha type (PRKCA) | Proto-oncogene c-RAF | RAF proto-oncogene serine/threonine-protein kinase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 76755.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The recombinant human PKC enzymes were
produced using a baculovirus expression system in SF9 cells |
|---|
| Residue: | 672 |
|---|
| Sequence: | MADVFPGNDSTASQDVANRFARKGALRQKNVHEVKDHKFIARFFKQPTFCSHCTDFIWGF
GKQGFQCQVCCFVVHKRCHEFVTFSCPGADKGPDTDDPRSKHKFKIHTYGSPTFCDHCGS
LLYGLIHQGMKCDTCDMNVHKQCVINVPSLCGMDHTEKRGRIYLKAEVADEKLHVTVRDA
KNLIPMDPNGLSDPYVKLKLIPDPKNESKQKTKTIRSTLNPQWNESFTFKLKPSDKDRRL
SVEIWDWDRTTRNDFMGSLSFGVSELMKMPASGWYKLLNQEEGEYYNVPIPEGDEEGNME
LRQKFEKAKLGPAGNKVISPSEDRKQPSNNLDRVKLTDFNFLMVLGKGSFGKVMLADRKG
TEELYAIKILKKDVVIQDDDVECTMVEKRVLALLDKPPFLTQLHSCFQTVDRLYFVMEYV
NGGDLMYHIQQVGKFKEPQAVFYAAEISIGLFFLHKRGIIYRDLKLDNVMLDSEGHIKIA
DFGMCKEHMMDGVTTRTFCGTPDYIAPEIIAYQPYGKSVDWWAYGVLLYEMLAGQPPFDG
EDEDELFQSIMEHNVSYPKSLSKEAVSVCKGLMTKHPAKRLGCGPEGERDVREHAFFRRI
DWEKLENREIQPPFKPKVCGKGAENFDKFFTRGQPVLTPPDQLVIANIDQSDFEGFSYVN
PQFVHPILQSAV
|
|
|
|---|
| BDBM50045370 |
|---|
| n/a |
|---|
| Name | BDBM50045370 |
|---|
| Synonyms: | CHEMBL3314140 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H21ClN4O |
|---|
| Mol. Mass. | 404.892 |
|---|
| SMILES | Clc1ccc(cc1)-c1ccc(o1)-c1nccn1-c1ccc(cc1)N1CCNCC1 |
|---|
| Structure | 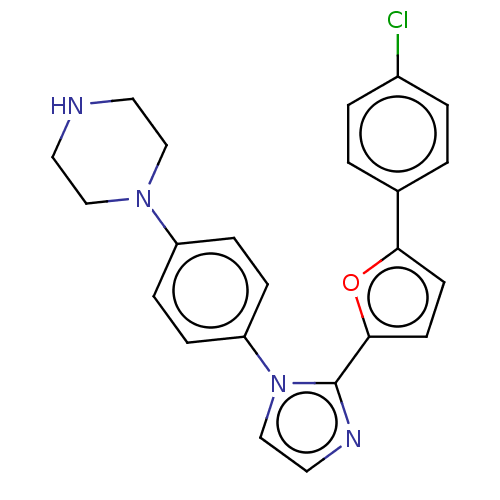
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xiao, D; Zhu, X; Sofolarides, M; Degrado, S; Shao, N; Rao, A; Chen, X; Aslanian, R; Fossetta, J; Tian, F; Trivedi, P; Lundell, D; Palani, A Discovery of a novel series of potent MK2 non-ATP competitive inhibitors using 1,2-substituted azoles as cis-amide isosteres. Bioorg Med Chem Lett24:3609-13 (2014) [PubMed] Article
Xiao, D; Zhu, X; Sofolarides, M; Degrado, S; Shao, N; Rao, A; Chen, X; Aslanian, R; Fossetta, J; Tian, F; Trivedi, P; Lundell, D; Palani, A Discovery of a novel series of potent MK2 non-ATP competitive inhibitors using 1,2-substituted azoles as cis-amide isosteres. Bioorg Med Chem Lett24:3609-13 (2014) [PubMed] Article