| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50051939 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1453047 (CHEMBL3362129) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Wang, HC; Jagtap, AD; Chang, PT; Liu, JR; Liu, CP; Tseng, HW; Chen, GS; Chern, JW Bioisosteric replacement of an acylureido moiety attached to an indolin-2-one scaffold with a malonamido or a 2/4-pyridinoylamido moiety produces a selectively potent Aurora-B inhibitor. Eur J Med Chem84:312-34 (2014) [PubMed] Article Wang, HC; Jagtap, AD; Chang, PT; Liu, JR; Liu, CP; Tseng, HW; Chen, GS; Chern, JW Bioisosteric replacement of an acylureido moiety attached to an indolin-2-one scaffold with a malonamido or a 2/4-pyridinoylamido moiety produces a selectively potent Aurora-B inhibitor. Eur J Med Chem84:312-34 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50051939 |
|---|
| n/a |
|---|
| Name | BDBM50051939 |
|---|
| Synonyms: | CHEMBL3322590 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H17FN4O3 |
|---|
| Mol. Mass. | 440.4259 |
|---|
| SMILES | Fc1ccccc1-c1c[nH]cc(C(=O)Nc2ccc3\C(=C\c4ccc[nH]4)C(=O)Nc3c2)c1=O |
|---|
| Structure | 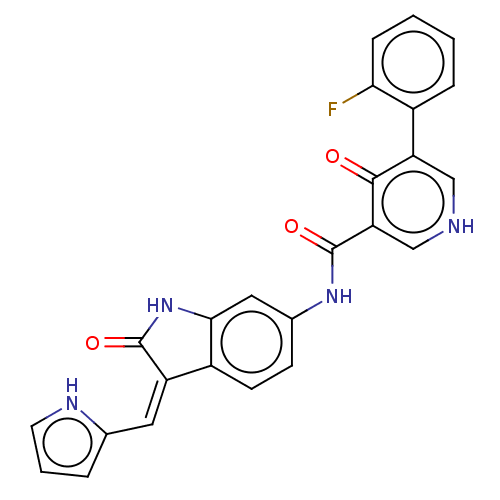
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, HC; Jagtap, AD; Chang, PT; Liu, JR; Liu, CP; Tseng, HW; Chen, GS; Chern, JW Bioisosteric replacement of an acylureido moiety attached to an indolin-2-one scaffold with a malonamido or a 2/4-pyridinoylamido moiety produces a selectively potent Aurora-B inhibitor. Eur J Med Chem84:312-34 (2014) [PubMed] Article
Wang, HC; Jagtap, AD; Chang, PT; Liu, JR; Liu, CP; Tseng, HW; Chen, GS; Chern, JW Bioisosteric replacement of an acylureido moiety attached to an indolin-2-one scaffold with a malonamido or a 2/4-pyridinoylamido moiety produces a selectively potent Aurora-B inhibitor. Eur J Med Chem84:312-34 (2014) [PubMed] Article