| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50054691 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1453127 (CHEMBL3364425) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Li, J; Kennedy, LJ; Wang, H; Li, JJ; Walker, SJ; Hong, Z; O'Connor, SP; Nayeem, A; Camac, DM; Morin, PE; Sheriff, S; Wang, M; Harper, T; Golla, R; Seethala, R; Harrity, T; Ponticiello, RP; Morgan, NN; Taylor, JR; Zebo, R; Gordon, DA; Robl, JA Optimization of 1,2,4-Triazolopyridines as Inhibitors of Human 11�-Hydroxysteroid Dehydrogenase Type 1 (11�-HSD-1). ACS Med Chem Lett5:803-8 (2014) [PubMed] Article Li, J; Kennedy, LJ; Wang, H; Li, JJ; Walker, SJ; Hong, Z; O'Connor, SP; Nayeem, A; Camac, DM; Morin, PE; Sheriff, S; Wang, M; Harper, T; Golla, R; Seethala, R; Harrity, T; Ponticiello, RP; Morgan, NN; Taylor, JR; Zebo, R; Gordon, DA; Robl, JA Optimization of 1,2,4-Triazolopyridines as Inhibitors of Human 11�-Hydroxysteroid Dehydrogenase Type 1 (11�-HSD-1). ACS Med Chem Lett5:803-8 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50054691 |
|---|
| n/a |
|---|
| Name | BDBM50054691 |
|---|
| Synonyms: | CHEMBL3318978 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H16ClN3 |
|---|
| Mol. Mass. | 309.793 |
|---|
| SMILES | Clc1ccc(cc1)C1(CC1)c1nnc2c(cccn12)C1CC1 |
|---|
| Structure | 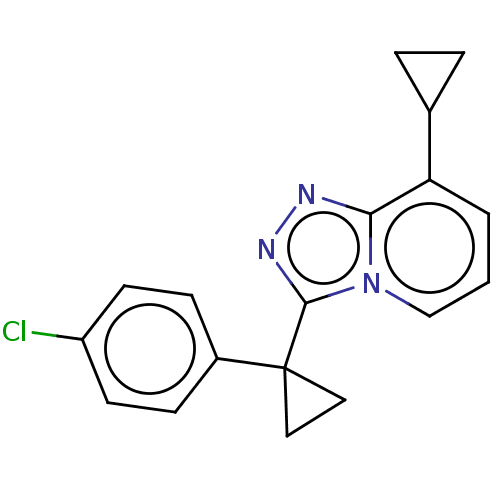
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, J; Kennedy, LJ; Wang, H; Li, JJ; Walker, SJ; Hong, Z; O'Connor, SP; Nayeem, A; Camac, DM; Morin, PE; Sheriff, S; Wang, M; Harper, T; Golla, R; Seethala, R; Harrity, T; Ponticiello, RP; Morgan, NN; Taylor, JR; Zebo, R; Gordon, DA; Robl, JA Optimization of 1,2,4-Triazolopyridines as Inhibitors of Human 11�-Hydroxysteroid Dehydrogenase Type 1 (11�-HSD-1). ACS Med Chem Lett5:803-8 (2014) [PubMed] Article
Li, J; Kennedy, LJ; Wang, H; Li, JJ; Walker, SJ; Hong, Z; O'Connor, SP; Nayeem, A; Camac, DM; Morin, PE; Sheriff, S; Wang, M; Harper, T; Golla, R; Seethala, R; Harrity, T; Ponticiello, RP; Morgan, NN; Taylor, JR; Zebo, R; Gordon, DA; Robl, JA Optimization of 1,2,4-Triazolopyridines as Inhibitors of Human 11�-Hydroxysteroid Dehydrogenase Type 1 (11�-HSD-1). ACS Med Chem Lett5:803-8 (2014) [PubMed] Article