| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D(3) dopamine receptor |
|---|
| Ligand | BDBM25771 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1449716 (CHEMBL3378139) |
|---|
| Ki | 1049±n/a nM |
|---|
| Citation |  Arnold, N; Beattie, D; Bradley, M; Brearley, A; Brown, L; Charlton, SJ; Fairhurst, RA; Farr, D; Fozard, J; Fullerton, J; Gosling, M; Hatto, J; Janus, D; Jones, D; Jordan, L; Lewis, C; Maas, J; McCarthy, C; Mercer, M; Oakman, H; Press, N; Profit, R; Schuerch, F; Sykes, D; Taylor, RJ; Trifilieff, A; Tuffnell, A The identification of 7-[(R)-2-((1S,2S)-2-benzyloxycyclopentylamino)-1-hydroxyethyl]-4-hydroxybenzothiazolone as an inhaled long-acting�2-adrenoceptor agonist. Bioorg Med Chem Lett24:4341-7 (2014) [PubMed] Article Arnold, N; Beattie, D; Bradley, M; Brearley, A; Brown, L; Charlton, SJ; Fairhurst, RA; Farr, D; Fozard, J; Fullerton, J; Gosling, M; Hatto, J; Janus, D; Jones, D; Jordan, L; Lewis, C; Maas, J; McCarthy, C; Mercer, M; Oakman, H; Press, N; Profit, R; Schuerch, F; Sykes, D; Taylor, RJ; Trifilieff, A; Tuffnell, A The identification of 7-[(R)-2-((1S,2S)-2-benzyloxycyclopentylamino)-1-hydroxyethyl]-4-hydroxybenzothiazolone as an inhaled long-acting�2-adrenoceptor agonist. Bioorg Med Chem Lett24:4341-7 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D(3) dopamine receptor |
|---|
| Name: | D(3) dopamine receptor |
|---|
| Synonyms: | DOPAMINE D3 | DRD3 | DRD3_HUMAN | Dopamine D3 receptor | Dopamine D3 receptor (D3) | Dopamine D3 receptor (D3R) | Dopamine receptor | Dopamine receptor (D3) | Dopamine receptor D3 |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 44243.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 400 |
|---|
| Sequence: | MASLSQLSSHLNYTCGAENSTGASQARPHAYYALSYCALILAIVFGNGLVCMAVLKERAL
QTTTNYLVVSLAVADLLVATLVMPWVVYLEVTGGVWNFSRICCDVFVTLDVMMCTASILN
LCAISIDRYTAVVMPVHYQHGTGQSSCRRVALMITAVWVLAFAVSCPLLFGFNTTGDPTV
CSISNPDFVIYSSVVSFYLPFGVTVLVYARIYVVLKQRRRKRILTRQNSQCNSVRPGFPQ
QTLSPDPAHLELKRYYSICQDTALGGPGFQERGGELKREEKTRNSLSPTIAPKLSLEVRK
LSNGRLSTSLKLGPLQPRGVPLREKKATQMVAIVLGAFIVCWLPFFLTHVLNTHCQTCHV
SPELYSATTWLGYVNSALNPVIYTTFNIEFRKAFLKILSC
|
|
|
|---|
| BDBM25771 |
|---|
| n/a |
|---|
| Name | BDBM25771 |
|---|
| Synonyms: | 1-hydroxy-2-naphthoic acid;4-[1-hydroxy-2-[6-(4-phenylbutoxy)hexylamino]ethyl]-2-methylol-phenol | 4-(1-hydroxy-2-{[6-(4-phenylbutoxy)hexyl]amino}ethyl)-2-(hydroxymethyl)phenol | Aeromax | Salmeterol | Serevent | salmeterol xinafoate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H37NO4 |
|---|
| Mol. Mass. | 415.5656 |
|---|
| SMILES | OCc1cc(ccc1O)C(O)CNCCCCCCOCCCCc1ccccc1 |
|---|
| Structure | 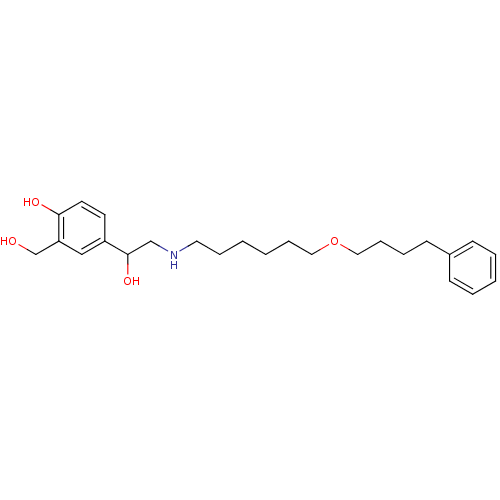
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Arnold, N; Beattie, D; Bradley, M; Brearley, A; Brown, L; Charlton, SJ; Fairhurst, RA; Farr, D; Fozard, J; Fullerton, J; Gosling, M; Hatto, J; Janus, D; Jones, D; Jordan, L; Lewis, C; Maas, J; McCarthy, C; Mercer, M; Oakman, H; Press, N; Profit, R; Schuerch, F; Sykes, D; Taylor, RJ; Trifilieff, A; Tuffnell, A The identification of 7-[(R)-2-((1S,2S)-2-benzyloxycyclopentylamino)-1-hydroxyethyl]-4-hydroxybenzothiazolone as an inhaled long-acting�2-adrenoceptor agonist. Bioorg Med Chem Lett24:4341-7 (2014) [PubMed] Article
Arnold, N; Beattie, D; Bradley, M; Brearley, A; Brown, L; Charlton, SJ; Fairhurst, RA; Farr, D; Fozard, J; Fullerton, J; Gosling, M; Hatto, J; Janus, D; Jones, D; Jordan, L; Lewis, C; Maas, J; McCarthy, C; Mercer, M; Oakman, H; Press, N; Profit, R; Schuerch, F; Sykes, D; Taylor, RJ; Trifilieff, A; Tuffnell, A The identification of 7-[(R)-2-((1S,2S)-2-benzyloxycyclopentylamino)-1-hydroxyethyl]-4-hydroxybenzothiazolone as an inhaled long-acting�2-adrenoceptor agonist. Bioorg Med Chem Lett24:4341-7 (2014) [PubMed] Article