| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50056386 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1451407 (CHEMBL3363967) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Voight, EA; Gomtsyan, AR; Daanen, JF; Perner, RJ; Schmidt, RG; Bayburt, EK; DiDomenico, S; McDonald, HA; Puttfarcken, PS; Chen, J; Neelands, TR; Bianchi, BR; Han, P; Reilly, RM; Franklin, PH; Segreti, JA; Nelson, RA; Su, Z; King, AJ; Polakowski, JS; Baker, SJ; Gauvin, DM; Lewis, LR; Mikusa, JP; Joshi, SK; Faltynek, CR; Kym, PR; Kort, ME Discovery of (R)-1-(7-chloro-2,2-bis(fluoromethyl)chroman-4-yl)-3-(3-methylisoquinolin-5-yl)urea (A-1165442): a temperature-neutral transient receptor potential vanilloid-1 (TRPV1) antagonist with analgesic efficacy. J Med Chem57:7412-24 (2014) [PubMed] Article Voight, EA; Gomtsyan, AR; Daanen, JF; Perner, RJ; Schmidt, RG; Bayburt, EK; DiDomenico, S; McDonald, HA; Puttfarcken, PS; Chen, J; Neelands, TR; Bianchi, BR; Han, P; Reilly, RM; Franklin, PH; Segreti, JA; Nelson, RA; Su, Z; King, AJ; Polakowski, JS; Baker, SJ; Gauvin, DM; Lewis, LR; Mikusa, JP; Joshi, SK; Faltynek, CR; Kym, PR; Kort, ME Discovery of (R)-1-(7-chloro-2,2-bis(fluoromethyl)chroman-4-yl)-3-(3-methylisoquinolin-5-yl)urea (A-1165442): a temperature-neutral transient receptor potential vanilloid-1 (TRPV1) antagonist with analgesic efficacy. J Med Chem57:7412-24 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50056386 |
|---|
| n/a |
|---|
| Name | BDBM50056386 |
|---|
| Synonyms: | CHEMBL3325460 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H22F3N3O2 |
|---|
| Mol. Mass. | 429.4349 |
|---|
| SMILES | Cc1cc2c(NC(=O)N[C@@H]3CC(C)(C)Oc4cc(ccc34)C(F)(F)F)cccc2cn1 |r| |
|---|
| Structure | 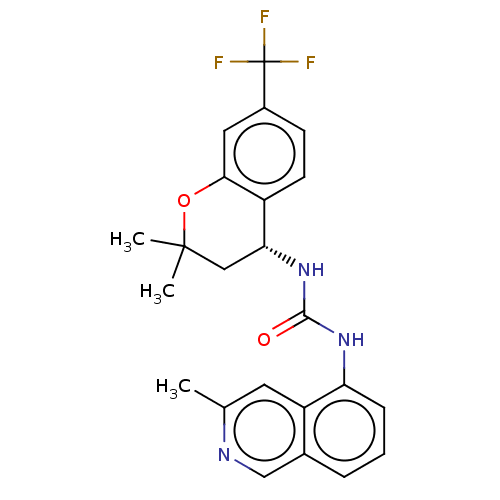
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Voight, EA; Gomtsyan, AR; Daanen, JF; Perner, RJ; Schmidt, RG; Bayburt, EK; DiDomenico, S; McDonald, HA; Puttfarcken, PS; Chen, J; Neelands, TR; Bianchi, BR; Han, P; Reilly, RM; Franklin, PH; Segreti, JA; Nelson, RA; Su, Z; King, AJ; Polakowski, JS; Baker, SJ; Gauvin, DM; Lewis, LR; Mikusa, JP; Joshi, SK; Faltynek, CR; Kym, PR; Kort, ME Discovery of (R)-1-(7-chloro-2,2-bis(fluoromethyl)chroman-4-yl)-3-(3-methylisoquinolin-5-yl)urea (A-1165442): a temperature-neutral transient receptor potential vanilloid-1 (TRPV1) antagonist with analgesic efficacy. J Med Chem57:7412-24 (2014) [PubMed] Article
Voight, EA; Gomtsyan, AR; Daanen, JF; Perner, RJ; Schmidt, RG; Bayburt, EK; DiDomenico, S; McDonald, HA; Puttfarcken, PS; Chen, J; Neelands, TR; Bianchi, BR; Han, P; Reilly, RM; Franklin, PH; Segreti, JA; Nelson, RA; Su, Z; King, AJ; Polakowski, JS; Baker, SJ; Gauvin, DM; Lewis, LR; Mikusa, JP; Joshi, SK; Faltynek, CR; Kym, PR; Kort, ME Discovery of (R)-1-(7-chloro-2,2-bis(fluoromethyl)chroman-4-yl)-3-(3-methylisoquinolin-5-yl)urea (A-1165442): a temperature-neutral transient receptor potential vanilloid-1 (TRPV1) antagonist with analgesic efficacy. J Med Chem57:7412-24 (2014) [PubMed] Article