| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Estrogen receptor |
|---|
| Ligand | BDBM50057651 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1448084 (CHEMBL3371989) |
|---|
| IC50 | >1000±n/a nM |
|---|
| Citation |  Ullrich, T; Sasmal, S; Boorgu, V; Pasagadi, S; Cheera, S; Rajagopalan, S; Bhumireddy, A; Shashikumar, D; Chelur, S; Belliappa, C; Pandit, C; Krishnamurthy, N; Mukherjee, S; Ramanathan, A; Ghadiyaram, C; Ramachandra, M; Santos, PG; Lagu, B; Bock, MG; Perrone, MH; Weiler, S; Keller, H 3-alkoxy-pyrrolo[1,2-b]pyrazolines as selective androgen receptor modulators with ideal physicochemical properties for transdermal administration. J Med Chem57:7396-411 (2014) [PubMed] Article Ullrich, T; Sasmal, S; Boorgu, V; Pasagadi, S; Cheera, S; Rajagopalan, S; Bhumireddy, A; Shashikumar, D; Chelur, S; Belliappa, C; Pandit, C; Krishnamurthy, N; Mukherjee, S; Ramanathan, A; Ghadiyaram, C; Ramachandra, M; Santos, PG; Lagu, B; Bock, MG; Perrone, MH; Weiler, S; Keller, H 3-alkoxy-pyrrolo[1,2-b]pyrazolines as selective androgen receptor modulators with ideal physicochemical properties for transdermal administration. J Med Chem57:7396-411 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Estrogen receptor |
|---|
| Name: | Estrogen receptor |
|---|
| Synonyms: | ER | ER-alpha | ESR | ESR1 | ESR1_HUMAN | Estradiol receptor | Estrogen receptor | Estrogen receptor (ER alpha) | Estrogen receptor (ER-alpha) | Estrogen receptor alpha (ER alpha) | Estrogen receptor alpha (ER) | NR3A1 | Nuclear receptor subfamily 3 group A member 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 66230.44 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P03372 |
|---|
| Residue: | 595 |
|---|
| Sequence: | MTMTLHTKASGMALLHQIQGNELEPLNRPQLKIPLERPLGEVYLDSSKPAVYNYPEGAAY
EFNAAAAANAQVYGQTGLPYGPGSEAAAFGSNGLGGFPPLNSVSPSPLMLLHPPPQLSPF
LQPHGQQVPYYLENEPSGYTVREAGPPAFYRPNSDNRRQGGRERLASTNDKGSMAMESAK
ETRYCAVCNDYASGYHYGVWSCEGCKAFFKRSIQGHNDYMCPATNQCTIDKNRRKSCQAC
RLRKCYEVGMMKGGIRKDRRGGRMLKHKRQRDDGEGRGEVGSAGDMRAANLWPSPLMIKR
SKKNSLALSLTADQMVSALLDAEPPILYSEYDPTRPFSEASMMGLLTNLADRELVHMINW
AKRVPGFVDLTLHDQVHLLECAWLEILMIGLVWRSMEHPGKLLFAPNLLLDRNQGKCVEG
MVEIFDMLLATSSRFRMMNLQGEEFVCLKSIILLNSGVYTFLSSTLKSLEEKDHIHRVLD
KITDTLIHLMAKAGLTLQQQHQRLAQLLLILSHIRHMSNKGMEHLYSMKCKNVVPLYDLL
LEMLDAHRLHAPTSRGGASVEETDQSHLATAGSTSSHSLQKYYITGEAEGFPATV
|
|
|
|---|
| BDBM50057651 |
|---|
| n/a |
|---|
| Name | BDBM50057651 |
|---|
| Synonyms: | CHEMBL3326454 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H16ClN3O2 |
|---|
| Mol. Mass. | 305.759 |
|---|
| SMILES | [H][C@@]12[C@@H](O)CCN1N=C([C@H]2OC)c1ccc(C#N)c(Cl)c1C |r,c:8| |
|---|
| Structure | 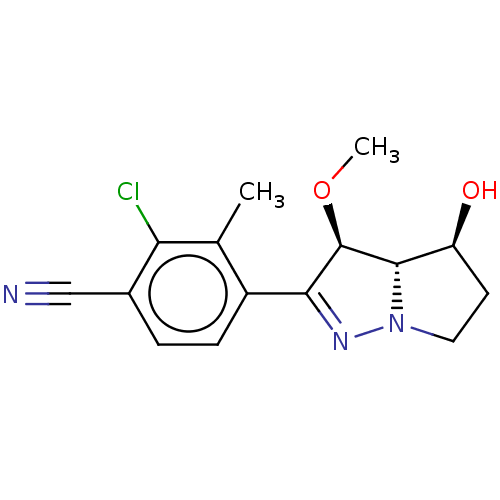
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ullrich, T; Sasmal, S; Boorgu, V; Pasagadi, S; Cheera, S; Rajagopalan, S; Bhumireddy, A; Shashikumar, D; Chelur, S; Belliappa, C; Pandit, C; Krishnamurthy, N; Mukherjee, S; Ramanathan, A; Ghadiyaram, C; Ramachandra, M; Santos, PG; Lagu, B; Bock, MG; Perrone, MH; Weiler, S; Keller, H 3-alkoxy-pyrrolo[1,2-b]pyrazolines as selective androgen receptor modulators with ideal physicochemical properties for transdermal administration. J Med Chem57:7396-411 (2014) [PubMed] Article
Ullrich, T; Sasmal, S; Boorgu, V; Pasagadi, S; Cheera, S; Rajagopalan, S; Bhumireddy, A; Shashikumar, D; Chelur, S; Belliappa, C; Pandit, C; Krishnamurthy, N; Mukherjee, S; Ramanathan, A; Ghadiyaram, C; Ramachandra, M; Santos, PG; Lagu, B; Bock, MG; Perrone, MH; Weiler, S; Keller, H 3-alkoxy-pyrrolo[1,2-b]pyrazolines as selective androgen receptor modulators with ideal physicochemical properties for transdermal administration. J Med Chem57:7396-411 (2014) [PubMed] Article