| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urokinase-type plasminogen activator |
|---|
| Ligand | BDBM50100897 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1525567 (CHEMBL3637193) |
|---|
| Ki | 5500±n/a nM |
|---|
| Citation |  Goswami, R; Wohlfahrt, G; T�rm�kangas, O; Moilanen, A; Lakshminarasimhan, A; Nagaraj, J; Arumugam, KN; Mukherjee, S; Chacko, AR; Krishnamurthy, NR; Jaleel, M; Palakurthy, RK; Samiulla, DS; Ramachandra, M Structure-guided discovery of 2-aryl/pyridin-2-yl-1H-indole derivatives as potent and selective hepsin inhibitors. Bioorg Med Chem Lett25:5309-14 (2015) [PubMed] Article Goswami, R; Wohlfahrt, G; T�rm�kangas, O; Moilanen, A; Lakshminarasimhan, A; Nagaraj, J; Arumugam, KN; Mukherjee, S; Chacko, AR; Krishnamurthy, NR; Jaleel, M; Palakurthy, RK; Samiulla, DS; Ramachandra, M Structure-guided discovery of 2-aryl/pyridin-2-yl-1H-indole derivatives as potent and selective hepsin inhibitors. Bioorg Med Chem Lett25:5309-14 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Urokinase-type plasminogen activator |
|---|
| Name: | Urokinase-type plasminogen activator |
|---|
| Synonyms: | 3.4.21.73 | PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator chain B | Urokinase-type plasminogen activator long chain A | Urokinase-type plasminogen activator short chain A | Urokinase-type plasminogen activator/surface receptor | uPA |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48528.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00749 |
|---|
| Residue: | 431 |
|---|
| Sequence: | MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQ
HCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHN
YCRNPDNRRRPWCYVQVGLKLLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKII
GGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLG
RSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICL
PSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKML
CAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIR
SHTKEENGLAL
|
|
|
|---|
| BDBM50100897 |
|---|
| n/a |
|---|
| Name | BDBM50100897 |
|---|
| Synonyms: | 2-(2-Hydroxy-phenyl)-1H-benzoimidazole-5-carboxamidine | 2-(2-Hydroxy-phenyl)-3H-benzoimidazole-5-carboxamidine | 2-{5-[AMINO(IMINIO)METHYL]-1H-BENZIMIDAZOL-2-YL}BENZENOLATE | CHEMBL433501 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H12N4O |
|---|
| Mol. Mass. | 252.2713 |
|---|
| SMILES | NC(=N)c1ccc2nc([nH]c2c1)-c1ccccc1O |
|---|
| Structure | 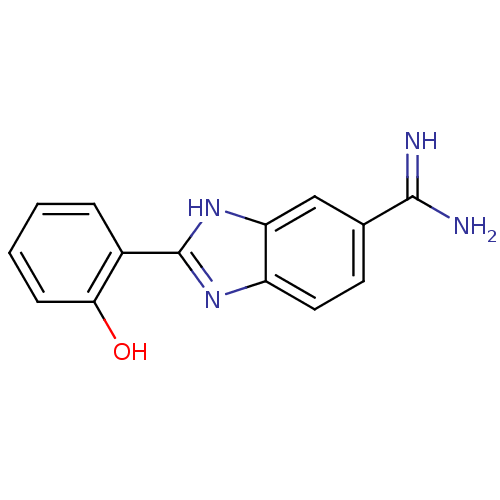
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Goswami, R; Wohlfahrt, G; T�rm�kangas, O; Moilanen, A; Lakshminarasimhan, A; Nagaraj, J; Arumugam, KN; Mukherjee, S; Chacko, AR; Krishnamurthy, NR; Jaleel, M; Palakurthy, RK; Samiulla, DS; Ramachandra, M Structure-guided discovery of 2-aryl/pyridin-2-yl-1H-indole derivatives as potent and selective hepsin inhibitors. Bioorg Med Chem Lett25:5309-14 (2015) [PubMed] Article
Goswami, R; Wohlfahrt, G; T�rm�kangas, O; Moilanen, A; Lakshminarasimhan, A; Nagaraj, J; Arumugam, KN; Mukherjee, S; Chacko, AR; Krishnamurthy, NR; Jaleel, M; Palakurthy, RK; Samiulla, DS; Ramachandra, M Structure-guided discovery of 2-aryl/pyridin-2-yl-1H-indole derivatives as potent and selective hepsin inhibitors. Bioorg Med Chem Lett25:5309-14 (2015) [PubMed] Article