| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin E synthase |
|---|
| Ligand | BDBM50140687 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1548641 (CHEMBL3757641) |
|---|
| IC50 | 3600±n/a nM |
|---|
| Citation |  Di Micco, S; Spatafora, C; Cardullo, N; Riccio, R; Fischer, K; Pergola, C; Koeberle, A; Werz, O; Chalal, M; Vervandier-Fasseur, D; Tringali, C; Bifulco, G 2,3-Dihydrobenzofuran privileged structures as new bioinspired lead compounds for the design of mPGES-1 inhibitors. Bioorg Med Chem24:820-6 (2016) [PubMed] Article Di Micco, S; Spatafora, C; Cardullo, N; Riccio, R; Fischer, K; Pergola, C; Koeberle, A; Werz, O; Chalal, M; Vervandier-Fasseur, D; Tringali, C; Bifulco, G 2,3-Dihydrobenzofuran privileged structures as new bioinspired lead compounds for the design of mPGES-1 inhibitors. Bioorg Med Chem24:820-6 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin E synthase |
|---|
| Name: | Prostaglandin E synthase |
|---|
| Synonyms: | MGST1L1 | MPGES1 | PGES | PIG12 | PTGES | PTGES_HUMAN | Prostaglandin E synthase (PGES-1) | Prostaglandin E synthase 1 (mPGES-1) | Prostaglandin E synthase-1 (PGES-1) | Prostaglandin E synthase/G/H synthase 2 | Prostaglandin E2 synthase-1 ( mPGES-1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 17112.22 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 152 |
|---|
| Sequence: | MPAHSLVMSSPALPAFLLCSTLLVIKMYVVAIITGQVRLRKKAFANPEDALRHGGPQYCR
SDPDVERCLRAHRNDMETIYPFLFLGFVYSFLGPNPFVAWMHFLVFLVGRVAHTVAYLGK
LRAPIRSVTYTLAQLPCASMALQILWEAARHL
|
|
|
|---|
| BDBM50140687 |
|---|
| n/a |
|---|
| Name | BDBM50140687 |
|---|
| Synonyms: | CHEMBL3752388 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H22O8 |
|---|
| Mol. Mass. | 414.4053 |
|---|
| SMILES | CCOC(=O)\C=C\c1cc2[C@H]([C@@H](Oc2c(O)c1)c1ccc(O)c(O)c1)C(=O)OCC |r| |
|---|
| Structure | 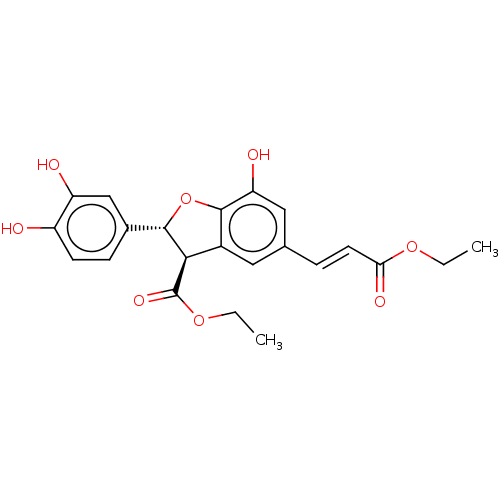
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Di Micco, S; Spatafora, C; Cardullo, N; Riccio, R; Fischer, K; Pergola, C; Koeberle, A; Werz, O; Chalal, M; Vervandier-Fasseur, D; Tringali, C; Bifulco, G 2,3-Dihydrobenzofuran privileged structures as new bioinspired lead compounds for the design of mPGES-1 inhibitors. Bioorg Med Chem24:820-6 (2016) [PubMed] Article
Di Micco, S; Spatafora, C; Cardullo, N; Riccio, R; Fischer, K; Pergola, C; Koeberle, A; Werz, O; Chalal, M; Vervandier-Fasseur, D; Tringali, C; Bifulco, G 2,3-Dihydrobenzofuran privileged structures as new bioinspired lead compounds for the design of mPGES-1 inhibitors. Bioorg Med Chem24:820-6 (2016) [PubMed] Article