| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50169351 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1577990 (CHEMBL3806689) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Wu, YJ; Guernon, J; Yang, F; Snyder, L; Shi, J; Mcclure, A; Rajamani, R; Park, H; Ng, A; Lewis, H; Chang, C; Camac, D; Toyn, JH; Ahlijanian, MK; Albright, CF; Macor, JE; Thompson, LA Targeting the BACE1 Active Site Flap Leads to a Potent Inhibitor That Elicits Robust Brain A� Reduction in Rodents. ACS Med Chem Lett7:271-6 (2016) [PubMed] Article Wu, YJ; Guernon, J; Yang, F; Snyder, L; Shi, J; Mcclure, A; Rajamani, R; Park, H; Ng, A; Lewis, H; Chang, C; Camac, D; Toyn, JH; Ahlijanian, MK; Albright, CF; Macor, JE; Thompson, LA Targeting the BACE1 Active Site Flap Leads to a Potent Inhibitor That Elicits Robust Brain A� Reduction in Rodents. ACS Med Chem Lett7:271-6 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50169351 |
|---|
| n/a |
|---|
| Name | BDBM50169351 |
|---|
| Synonyms: | CHEMBL3805852 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H22F2N6O3S |
|---|
| Mol. Mass. | 488.51 |
|---|
| SMILES | COc1cnc(cn1)C(=O)Nc1cc(F)c(F)c(c1)[C@]1(C)C[C@H](SC(N)=N1)c1c(C)noc1C |r,c:27| |
|---|
| Structure | 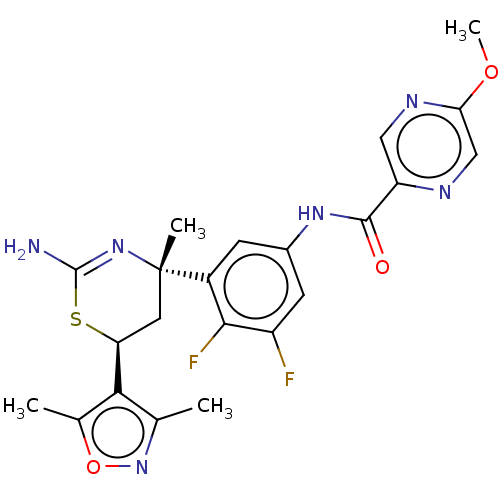
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wu, YJ; Guernon, J; Yang, F; Snyder, L; Shi, J; Mcclure, A; Rajamani, R; Park, H; Ng, A; Lewis, H; Chang, C; Camac, D; Toyn, JH; Ahlijanian, MK; Albright, CF; Macor, JE; Thompson, LA Targeting the BACE1 Active Site Flap Leads to a Potent Inhibitor That Elicits Robust Brain A� Reduction in Rodents. ACS Med Chem Lett7:271-6 (2016) [PubMed] Article
Wu, YJ; Guernon, J; Yang, F; Snyder, L; Shi, J; Mcclure, A; Rajamani, R; Park, H; Ng, A; Lewis, H; Chang, C; Camac, D; Toyn, JH; Ahlijanian, MK; Albright, CF; Macor, JE; Thompson, LA Targeting the BACE1 Active Site Flap Leads to a Potent Inhibitor That Elicits Robust Brain A� Reduction in Rodents. ACS Med Chem Lett7:271-6 (2016) [PubMed] Article