| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetyl-CoA carboxylase 2 |
|---|
| Ligand | BDBM50198329 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1620629 |
|---|
| IC50 | 290±n/a nM |
|---|
| Citation |  Okazaki, S; Noguchi-Yachide, T; Sakai, T; Ishikawa, M; Makishima, M; Hashimoto, Y; Yamaguchi, T Discovery of N-(1-(3-(4-phenoxyphenyl)-1,2,4-oxadiazol-5-yl)ethyl)acetamides as novel acetyl-CoA carboxylase 2 (ACC2) inhibitors with peroxisome proliferator-activated receptora/d (PPARa/d) dual agonistic activity. Bioorg Med Chem24:5258-5269 (2016) [PubMed] Article Okazaki, S; Noguchi-Yachide, T; Sakai, T; Ishikawa, M; Makishima, M; Hashimoto, Y; Yamaguchi, T Discovery of N-(1-(3-(4-phenoxyphenyl)-1,2,4-oxadiazol-5-yl)ethyl)acetamides as novel acetyl-CoA carboxylase 2 (ACC2) inhibitors with peroxisome proliferator-activated receptora/d (PPARa/d) dual agonistic activity. Bioorg Med Chem24:5258-5269 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetyl-CoA carboxylase 2 |
|---|
| Name: | Acetyl-CoA carboxylase 2 |
|---|
| Synonyms: | ACACB | ACACB_HUMAN | ACC-beta | ACC2 | ACCB | Acetyl-CoA carboxylase | Acetyl-CoA carboxylase 2 | Acetyl-CoA carboxylase 2 (ACC) | Acetyl-CoA carboxylase 2 (ACC2) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 276535.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O00763 |
|---|
| Residue: | 2458 |
|---|
| Sequence: | MVLLLCLSCLIFSCLTFSWLKIWGKMTDSKPITKSKSEANLIPSQEPFPASDNSGETPQR
NGEGHTLPKTPSQAEPASHKGPKDAGRRRNSLPPSHQKPPRNPLSSSDAAPSPELQANGT
GTQGLEATDTNGLSSSARPQGQQAGSPSKEDKKQANIKRQLMTNFILGSFDDYSSDEDSV
AGSSRESTRKGSRASLGALSLEAYLTTGEAETRVPTMRPSMSGLHLVKRGREHKKLDLHR
DFTVASPAEFVTRFGGDRVIEKVLIANNGIAAVKCMRSIRRWAYEMFRNERAIRFVVMVT
PEDLKANAEYIKMADHYVPVPGGPNNNNYANVELIVDIAKRIPVQAVWAGWGHASENPKL
PELLCKNGVAFLGPPSEAMWALGDKIASTVVAQTLQVPTLPWSGSGLTVEWTEDDLQQGK
RISVPEDVYDKGCVKDVDEGLEAAERIGFPLMIKASEGGGGKGIRKAESAEDFPILFRQV
QSEIPGSPIFLMKLAQHARHLEVQILADQYGNAVSLFGRDCSIQRRHQKIVEEAPATIAP
LAIFEFMEQCAIRLAKTVGYVSAGTVEYLYSQDGSFHFLELNPRLQVEHPCTEMIADVNL
PAAQLQIAMGVPLHRLKDIRLLYGESPWGVTPISFETPSNPPLARGHVIAARITSENPDE
GFKPSSGTVQELNFRSSKNVWGYFSVAATGGLHEFADSQFGHCFSWGENREEAISNMVVA
LKELSIRGDFRTTVEYLINLLETESFQNNDIDTGWLDYLIAEKVQAEKPDIMLGVVCGAL
NVADAMFRTCMTDFLHSLERGQVLPADSLLNLVDVELIYGGVKYILKVARQSLTMFVLIM
NGCHIEIDAHRLNDGGLLLSYNGNSYTTYMKEEVDSYRITIGNKTCVFEKENDPTVLRSP
SAGKLTQYTVEDGGHVEAGSSYAEMEVMKMIMTLNVQERGRVKYIKRPGAVLEAGCVVAR
LELDDPSKVHPAEPFTGELPAQQTLPILGEKLHQVFHSVLENLTNVMSGFCLPEPVFSIK
LKEWVQKLMMTLRHPSLPLLELQEIMTSVAGRIPAPVEKSVRRVMAQYASNITSVLCQFP
SQQIATILDCHAATLQRKADREVFFINTQSIVQLVQRYRSGIRGYMKTVVLDLLRRYLRV
EHHFQQAHYDKCVINLREQFKPDMSQVLDCIFSHAQVAKKNQLVIMLIDELCGPDPSLSD
ELISILNELTQLSKSEHCKVALRARQILIASHLPSYELRHNQVESIFLSAIDMYGHQFCP
ENLKKLILSETTIFDVLPTFFYHANKVVCMASLEVYVRRGYIAYELNSLQHRQLPDGTCV
VEFQFMLPSSHPNRMTVPISITNPDLLRHSTELFMDSGFSPLCQRMGAMVAFRRFEDFTR
NFDEVISCFANVPKDTPLFSEARTSLYSEDDCKSLREEPIHILNVSIQCADHLEDEALVP
ILRTFVQSKKNILVDYGLRRITFLIAQEKEFPKFFTFRARDEFAEDRIYRHLEPALAFQL
ELNRMRNFDLTAVPCANHKMHLYLGAAKVKEGVEVTDHRFFIRAIIRHSDLITKEASFEY
LQNEGERLLLEAMDELEVAFNNTSVRTDCNHIFLNFVPTVIMDPFKIEESVRYMVMRYGS
RLWKLRVLQAEVKINIRQTTTGSAVPIRLFITNESGYYLDISLYKEVTDSRSGNIMFHSF
GNKQGPQHGMLINTPYVTKDLLQAKRFQAQTLGTTYIYDFPEMFRQALFKLWGSPDKYPK
DILTYTELVLDSQGQLVEMNRLPGGNEVGMVAFKMRFKTQEYPEGRDVIVIGNDITFRIG
SFGPGEDLLYLRASEMARAEGIPKIYVAANSGARIGMAEEIKHMFHVAWVDPEDPHKGFK
YLYLTPQDYTRISSLNSVHCKHIEEGGESRYMITDIIGKDDGLGVENLRGSGMIAGESSL
AYEEIVTISLVTCRAIGIGAYLVRLGQRVIQVENSHIILTGASALNKVLGREVYTSNNQL
GGVQIMHYNGVSHITVPDDFEGVYTILEWLSYMPKDNHSPVPIITPTDPIDREIEFLPSR
APYDPRWMLAGRPHPTLKGTWQSGFFDHGSFKEIMAPWAQTVVTGRARLGGIPVGVIAVE
TRTVEVAVPADPANLDSEAKIIQQAGQVWFPDSAYKTAQAVKDFNREKLPLMIFANWRGF
SGGMKDMYDQVLKFGAYIVDGLRQYKQPILIYIPPYAELRGGSWVVIDATINPLCIEMYA
DKESRGGVLEPEGTVEIKFRKKDLIKSMRRIDPAYKKLMEQLGEPDLSDKDRKDLEGRLK
AREDLLLPIYHQVAVQFADFHDTPGRMLEKGVISDILEWKTARTFLYWRLRRLLLEDQVK
QEILQASGELSHVHIQSMLRRWFVETEGAVKAYLWDNNQVVVQWLEQHWQAGDGPRSTIR
ENITYLKHDSVLKTIRGLVEENPEVAVDCVIYLSQHISPAERAQVVHLLSTMDSPAST
|
|
|
|---|
| BDBM50198329 |
|---|
| n/a |
|---|
| Name | BDBM50198329 |
|---|
| Synonyms: | CHEMBL3981588 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H17N3O3 |
|---|
| Mol. Mass. | 323.3459 |
|---|
| SMILES | CC(NC(C)=O)c1nc(no1)-c1ccc(Oc2ccccc2)cc1 |
|---|
| Structure | 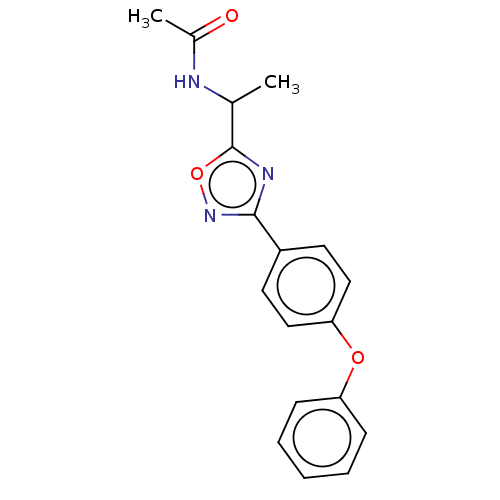
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Okazaki, S; Noguchi-Yachide, T; Sakai, T; Ishikawa, M; Makishima, M; Hashimoto, Y; Yamaguchi, T Discovery of N-(1-(3-(4-phenoxyphenyl)-1,2,4-oxadiazol-5-yl)ethyl)acetamides as novel acetyl-CoA carboxylase 2 (ACC2) inhibitors with peroxisome proliferator-activated receptora/d (PPARa/d) dual agonistic activity. Bioorg Med Chem24:5258-5269 (2016) [PubMed] Article
Okazaki, S; Noguchi-Yachide, T; Sakai, T; Ishikawa, M; Makishima, M; Hashimoto, Y; Yamaguchi, T Discovery of N-(1-(3-(4-phenoxyphenyl)-1,2,4-oxadiazol-5-yl)ethyl)acetamides as novel acetyl-CoA carboxylase 2 (ACC2) inhibitors with peroxisome proliferator-activated receptora/d (PPARa/d) dual agonistic activity. Bioorg Med Chem24:5258-5269 (2016) [PubMed] Article