| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor XI |
|---|
| Ligand | BDBM285898 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Factor XIa Assay |
|---|
| pH | 7.4±n/a |
|---|
| IC50 | 0.330±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  Mertz, E; Edmondson, SD; So, S; Sun, W; Liu, W; Neelamkavil, SF; Gao, Y; Hruza, A; Zang, Y; Ali, A; Mal, R; He, J; Kuang, R; Wu, H; Ogawa, AK; Nolting, AF Factor XIa inhibitors US Patent US10081617 Publication Date 9/25/2018 Mertz, E; Edmondson, SD; So, S; Sun, W; Liu, W; Neelamkavil, SF; Gao, Y; Hruza, A; Zang, Y; Ali, A; Mal, R; He, J; Kuang, R; Wu, H; Ogawa, AK; Nolting, AF Factor XIa inhibitors US Patent US10081617 Publication Date 9/25/2018 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor XI |
|---|
| Name: | Coagulation factor XI |
|---|
| Synonyms: | Coagulation factor XIa | Coagulation factor XIa heavy chain | Coagulation factor XIa light chain | F11 | FA11_HUMAN | FXI | Factor XIa | Factor XIa (fXIa) | PTA | Plasma thromboplastin antecedent |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 70130.58 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P03951 |
|---|
| Residue: | 625 |
|---|
| Sequence: | MIFLYQVVHFILFTSVSGECVTQLLKDTCFEGGDITTVFTPSAKYCQVVCTYHPRCLLFT
FTAESPSEDPTRWFTCVLKDSVTETLPRVNRTAAISGYSFKQCSHQISACNKDIYVDLDM
KGINYNSSVAKSAQECQERCTDDVHCHFFTYATRQFPSLEHRNICLLKHTQTGTPTRITK
LDKVVSGFSLKSCALSNLACIRDIFPNTVFADSNIDSVMAPDAFVCGRICTHHPGCLFFT
FFSQEWPKESQRNLCLLKTSESGLPSTRIKKSKALSGFSLQSCRHSIPVFCHSSFYHDTD
FLGEELDIVAAKSHEACQKLCTNAVRCQFFTYTPAQASCNEGKGKCYLKLSSNGSPTKIL
HGRGGISGYTLRLCKMDNECTTKIKPRIVGGTASVRGEWPWQVTLHTTSPTQRHLCGGSI
IGNQWILTAAHCFYGVESPKILRVYSGILNQSEIKEDTSFFGVQEIIIHDQYKMAESGYD
IALLKLETTVNYTDSQRPICLPSKGDRNVIYTDCWVTGWGYRKLRDKIQNTLQKAKIPLV
TNEECQKRYRGHKITHKMICAGYREGGKDACKGDSGGPLSCKHNEVWHLVGITSWGEGCA
QRERPGVYTNVVEYVDWILEKTQAV
|
|
|
|---|
| BDBM285898 |
|---|
| n/a |
|---|
| Name | BDBM285898 |
|---|
| Synonyms: | 7-(5-(5-carboxythiophen-2-yl)-4-chloro-1H-imidazol-2-yl)-3-(5-chloro-2-(1H-tetrazol-1-yl)phenyl)-7-hydroxy-6,7-dihydro-5H-cyclopenta[b]pyridine | US10081617, Example 46 | US10081617, Example 47 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H32Cl2N4O6S |
|---|
| Mol. Mass. | 659.581 |
|---|
| SMILES | CC(C)(C)OC(=O)Nc1ccc(Cl)cc1C1=CC2=C([N]([O-])=C1)C(O)(CC2)c1nc(Cl)c([nH]1)-c1ccc(s1)C(=O)OC(C)(C)C |c:18,21,t:16| |
|---|
| Structure | 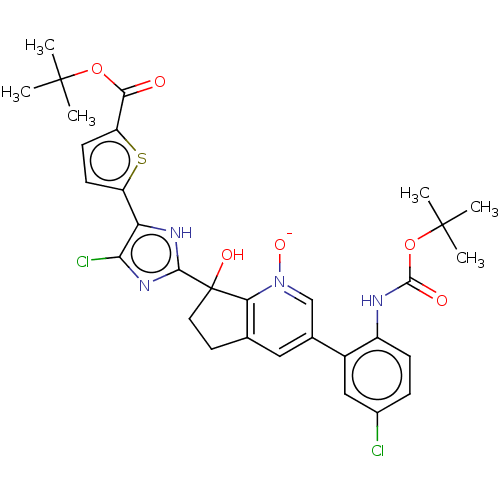
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mertz, E; Edmondson, SD; So, S; Sun, W; Liu, W; Neelamkavil, SF; Gao, Y; Hruza, A; Zang, Y; Ali, A; Mal, R; He, J; Kuang, R; Wu, H; Ogawa, AK; Nolting, AF Factor XIa inhibitors US Patent US10081617 Publication Date 9/25/2018
Mertz, E; Edmondson, SD; So, S; Sun, W; Liu, W; Neelamkavil, SF; Gao, Y; Hruza, A; Zang, Y; Ali, A; Mal, R; He, J; Kuang, R; Wu, H; Ogawa, AK; Nolting, AF Factor XIa inhibitors US Patent US10081617 Publication Date 9/25/2018