| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carboxylic ester hydrolase |
|---|
| Ligand | BDBM227585 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | In Vitro AChE/BChE Inhibition Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| IC50 | 4.76±0.58 nM |
|---|
| Comments | extracted |
|---|
| Citation |  Czarnecka, K; Szymanski, P; Girek, M; Mikiciuk-Olasik, E; Skibinski, R; Kabzinski, J; Majsterek, I; Malawska, B; Jonczyk, J; Bajda, M Tetrahydroacridine derivatives with fluorobenzoic acid moiety as multifunctional agents for Alzheimer's disease treatment. Bioorg Chem72:315-322 (2017) [PubMed] Article Czarnecka, K; Szymanski, P; Girek, M; Mikiciuk-Olasik, E; Skibinski, R; Kabzinski, J; Majsterek, I; Malawska, B; Jonczyk, J; Bajda, M Tetrahydroacridine derivatives with fluorobenzoic acid moiety as multifunctional agents for Alzheimer's disease treatment. Bioorg Chem72:315-322 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carboxylic ester hydrolase |
|---|
| Name: | Carboxylic ester hydrolase |
|---|
| Synonyms: | BuChE | Butyrlcholinesterase (BuChE) | Butyrylcholine esterase | Butyrylcholinesterase | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | Butyrylcholinesterase (EqBuChE) | Carboxylic ester hydrolase | butyrylcholinesterase precursor |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 68842.83 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | Q9N1N9 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MQSWGTIICIRILLRFLLLWVLIGNSHTEEDIIITTKNGKVRGMNLPVLGGTVTAFLGIP
YAQPPLGRLRFKKPQSLTKWSNIWNATKYANSCYQNTDQSFPGFLGSEMWNPNTELSEDC
LYLNVWIPAPKPKNATVMIWIYGGGFQTGTSSLPVYDGKFLARVERVIVVSMNYRVGALG
FLALSENPEAPGNMGLFDQQLALQWVQKNIAAFGGNPRSVTLFGESAGAASVSLHLLSPR
SQPLFTRAILQSGSSNAPWAVTSLYEARNRTLTLAKRMGCSRDNETEMIKCLRDKDPQEI
LLNEVFVVPYDTLLSVNFGPTVDGDFLTDMPDTLLQLGQFKRTQILVGVNKDEGTAFLVY
GAPGFSKDNNSIITRKEFQEGLKIFFPRVSEFGRESILFHYMDWLDDQRAENYREALDDV
VGDYNIICPALEFTKKFSELGNDAFFYYFEHRSTKLPWPEWMGVMHGYEIEFVFGLPLER
RVNYTKAEEILSRSIMKRWANFAKYGNPNGTQSNSTRWPVFKSTEQKYLTLNTESPKVYT
KLRAQQCRFWTLFFPKVLELTGNIDEAEREWKAGFHRWNNYMMDWKNQFNDYTSKKESCS
DF
|
|
|
|---|
| BDBM227585 |
|---|
| n/a |
|---|
| Name | BDBM227585 |
|---|
| Synonyms: | 3-Fluoro-N-[2-(1,2,3,4-tetrahydroacridin-9-ylamino)ethyl]benzamide hydrochloride (3d) |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H22FN3O |
|---|
| Mol. Mass. | 363.428 |
|---|
| SMILES | Fc1cccc(c1)C(=O)NCCNc1c2CCCCc2nc2ccccc12 |
|---|
| Structure | 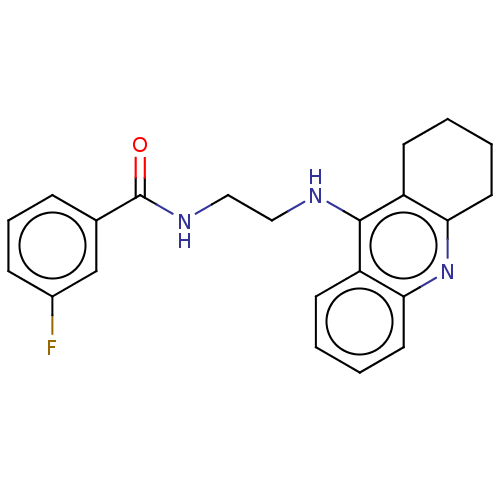
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Czarnecka, K; Szymanski, P; Girek, M; Mikiciuk-Olasik, E; Skibinski, R; Kabzinski, J; Majsterek, I; Malawska, B; Jonczyk, J; Bajda, M Tetrahydroacridine derivatives with fluorobenzoic acid moiety as multifunctional agents for Alzheimer's disease treatment. Bioorg Chem72:315-322 (2017) [PubMed] Article
Czarnecka, K; Szymanski, P; Girek, M; Mikiciuk-Olasik, E; Skibinski, R; Kabzinski, J; Majsterek, I; Malawska, B; Jonczyk, J; Bajda, M Tetrahydroacridine derivatives with fluorobenzoic acid moiety as multifunctional agents for Alzheimer's disease treatment. Bioorg Chem72:315-322 (2017) [PubMed] Article