| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase, H3 lysine-79 specific |
|---|
| Ligand | BDBM297394 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Assays of Enzymatic Activity |
|---|
| Ki | 13.0±7 nM |
|---|
| Kd | 1.7±0.2 nM |
|---|
| KOFF | 0.02 s-1 |
|---|
| KON | 12000000 M-1s-1 |
|---|
| Citation |  Olhava, EJ; Chesworth, R; Pollock, RM; Jin, L Inhibitors of protein methyltransferase DOT1L and methods of use thereof US Patent US10112968 Publication Date 10/30/2018 Olhava, EJ; Chesworth, R; Pollock, RM; Jin, L Inhibitors of protein methyltransferase DOT1L and methods of use thereof US Patent US10112968 Publication Date 10/30/2018 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase, H3 lysine-79 specific |
|---|
| Name: | Histone-lysine N-methyltransferase, H3 lysine-79 specific |
|---|
| Synonyms: | 2.1.1.43 | DOT1-like protein | DOT1-like protein (Dot1L) | DOT1L | DOT1L_HUMAN | H3-K79-HMTase | Histone H3-K79 methyltransferase | Histone H3-K79 methyltransferase (DOT1L) | Histone Methyltransferase DOT1L | Histone-lysine N-methyltransferase, H3 lysine-79 specific (DOT1L) | KIAA1814 | KMT4 | Lysine N-methyltransferase 4 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 184911.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q8TEK3 |
|---|
| Residue: | 1537 |
|---|
| Sequence: | MGEKLELRLKSPVGAEPAVYPWPLPVYDKHHDAAHEIIETIRWVCEEIPDLKLAMENYVL
IDYDTKSFESMQRLCDKYNRAIDSIHQLWKGTTQPMKLNTRPSTGLLRHILQQVYNHSVT
DPEKLNNYEPFSPEVYGETSFDLVAQMIDEIKMTDDDLFVDLGSGVGQVVLQVAAATNCK
HHYGVEKADIPAKYAETMDREFRKWMKWYGKKHAEYTLERGDFLSEEWRERIANTSVIFV
NNFAFGPEVDHQLKERFANMKEGGRIVSSKPFAPLNFRINSRNLSDIGTIMRVVELSPLK
GSVSWTGKPVSYYLHTIDRTILENYFSSLKNPKLREEQEAARRRQQRESKSNAATPTKGP
EGKVAGPADAPMDSGAEEEKAGAATVKKPSPSKARKKKLNKKGRKMAGRKRGRPKKMNTA
NPERKPKKNQTALDALHAQTVSQTAASSPQDAYRSPHSPFYQLPPSVQRHSPNPLLVAPT
PPALQKLLESFKIQYLQFLAYTKTPQYKASLQELLGQEKEKNAQLLGAAQQLLSHCQAQK
EEIRRLFQQKLDELGVKALTYNDLIQAQKEISAHNQQLREQSEQLEQDNRALRGQSLQLL
KARCEELQLDWATLSLEKLLKEKQALKSQISEKQRHCLELQISIVELEKSQRQQELLQLK
SCVPPDDALSLHLRGKGALGRELEPDASRLHLELDCTKFSLPHLSSMSPELSMNGQAAGY
ELCGVLSRPSSKQNTPQYLASPLDQEVVPCTPSHVGRPRLEKLSGLAAPDYTRLSPAKIV
LRRHLSQDHTVPGRPAASELHSRAEHTKENGLPYQSPSVPGSMKLSPQDPRPLSPGALQL
AGEKSSEKGLRERAYGSSGELITSLPISIPLSTVQPNKLPVSIPLASVVLPSRAERARST
PSPVLQPRDPSSTLEKQIGANAHGAGSRSLALAPAGFSYAGSVAISGALAGSPASLTPGA
EPATLDESSSSGSLFATVGSRSSTPQHPLLLAQPRNSLPASPAHQLSSSPRLGGAAQGPL
PEASKGDLPSDSGFSDPESEAKRRIVFTITTGAGSAKQSPSSKHSPLTASARGDCVPSHG
QDSRRRGRRKRASAGTPSLSAGVSPKRRALPSVAGLFTQPSGSPLNLNSMVSNINQPLEI
TAISSPETSLKSSPVPYQDHDQPPVLKKERPLSQTNGAHYSPLTSDEEPGSEDEPSSARI
ERKIATISLESKSPPKTLENGGGLAGRKPAPAGEPVNSSKWKSTFSPISDIGLAKSADSP
LQASSALSQNSLFTFRPALEEPSADAKLAAHPRKGFPGSLSGADGLSPGTNPANGCTFGG
GLAADLSLHSFSDGASLPHKGPEAAGLSSPLSFPSQRGKEGSDANPFLSKRQLDGLAGLK
GEGSRGKEAGEGGLPLCGPTDKTPLLSGKAAKARDREVDLKNGHNLFISAAAVPPGSLLS
GPGLAPAASSAGGAASSAQTHRSFLGPFPPGPQFALGPMSLQANLGSVAGSSVLQSLFSS
VPAAAGLVHVSSAATRLTNSHAMGSFSGVAGGTVGGN
|
|
|
|---|
| BDBM297394 |
|---|
| n/a |
|---|
| Name | BDBM297394 |
|---|
| Synonyms: | US10112968, Compound C118 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H36N8O4 |
|---|
| Mol. Mass. | 512.6045 |
|---|
| SMILES | CN(CCCNC(=O)Nc1ccc(cc1)C(C)(C)C)C[C@H]1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| |
|---|
| Structure | 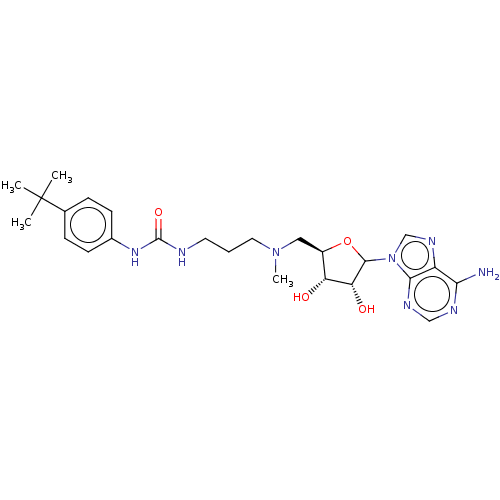
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Olhava, EJ; Chesworth, R; Pollock, RM; Jin, L Inhibitors of protein methyltransferase DOT1L and methods of use thereof US Patent US10112968 Publication Date 10/30/2018
Olhava, EJ; Chesworth, R; Pollock, RM; Jin, L Inhibitors of protein methyltransferase DOT1L and methods of use thereof US Patent US10112968 Publication Date 10/30/2018