

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM104110 3-Hydroxy-N-[(1S,2R)-2-hydroxy-3-[[[5-(5-isoxazolyl)-2-thienyl]sulfonyl](2- methylbutyl)amino]-1-(phenylmethyl)propyl]-benzamide (AF-80)
SMILES: CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1cccc(O)c1)S(=O)(=O)c1ccc(s1)-c1ccno1
InChI Key: InChIKey=YMMHVVLAGFSMTG-BJKOFHAPSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HIV-1 protease M2 (Human immunodeficiency virus) | BDBM104110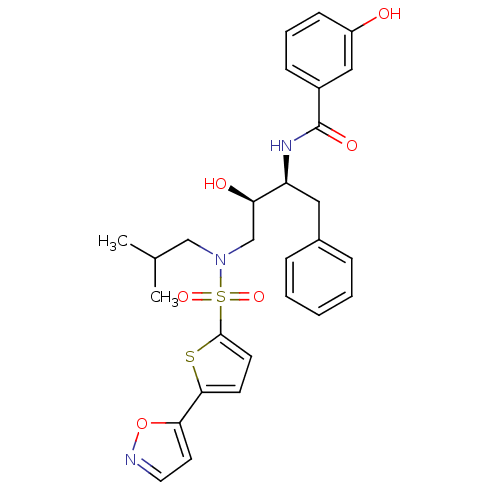 (3-Hydroxy-N-[(1S,2R)-2-hydroxy-3-[[[5-(5-isoxazoly...) | PDB MMDB B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Massachusetts Institute of Technology | Assay Description HIV protease inhibitor activities were determined by fluorescence resonance energy transfer (FRET) method. Protease substrate, Arg-Glu-(EDANS)-Ser-G... | ACS Chem Biol 8: 2433-41 (2013) Article DOI: 10.1021/cb400468c BindingDB Entry DOI: 10.7270/Q2R2101Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease M3 (Human immunodeficiency virus) | BDBM104110 (3-Hydroxy-N-[(1S,2R)-2-hydroxy-3-[[[5-(5-isoxazoly...) | PDB MMDB B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 10.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Massachusetts Institute of Technology | Assay Description HIV protease inhibitor activities were determined by fluorescence resonance energy transfer (FRET) method. Protease substrate, Arg-Glu-(EDANS)-Ser-G... | ACS Chem Biol 8: 2433-41 (2013) Article DOI: 10.1021/cb400468c BindingDB Entry DOI: 10.7270/Q2R2101Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease M4 (Human immunodeficiency virus) | BDBM104110 (3-Hydroxy-N-[(1S,2R)-2-hydroxy-3-[[[5-(5-isoxazoly...) | PDB MMDB B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 13.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Massachusetts Institute of Technology | Assay Description HIV protease inhibitor activities were determined by fluorescence resonance energy transfer (FRET) method. Protease substrate, Arg-Glu-(EDANS)-Ser-G... | ACS Chem Biol 8: 2433-41 (2013) Article DOI: 10.1021/cb400468c BindingDB Entry DOI: 10.7270/Q2R2101Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease M1 (Human immunodeficiency virus) | BDBM104110 (3-Hydroxy-N-[(1S,2R)-2-hydroxy-3-[[[5-(5-isoxazoly...) | PDB MMDB B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 28.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Massachusetts Institute of Technology | Assay Description HIV protease inhibitor activities were determined by fluorescence resonance energy transfer (FRET) method. Protease substrate, Arg-Glu-(EDANS)-Ser-G... | ACS Chem Biol 8: 2433-41 (2013) Article DOI: 10.1021/cb400468c BindingDB Entry DOI: 10.7270/Q2R2101Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||