

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM13054 (2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H-1,7-thieno[2,3-b]thiopyran-6-sulfonamide::Dorzolamide, DZA::sulfonamide 5
SMILES: CCN[C@@H]1C[C@@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O
InChI Key: InChIKey=IAVUPMFITXYVAF-HTRCEHHLSA-N
Data: 13 KI
PDB links: 1 PDB ID contains this monomer as substructures. 1 PDB ID contains inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM13054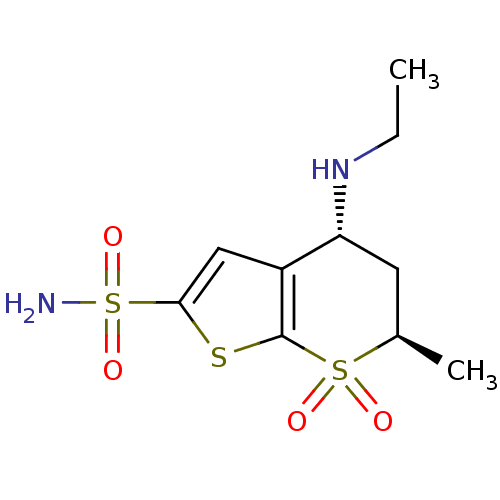 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 9 | -10.8 | n/a | n/a | n/a | n/a | n/a | 7.5 | 20 |
Institut des Biomolécules Max Mousseron (IBMM) | Assay Description Inhibition assay using carbonic anhydrases with an SX.18MV-R, a stopped-flow instrument from Applied Photophysics was used for assaying the CA cataly... | Chem Biol Drug Des 74: 636-9 (2009) Article DOI: 10.1111/j.1747-0285.2009.00902.x BindingDB Entry DOI: 10.7270/Q2VX0F1H | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 9 | -10.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| Carbonic anhydrase 6 (CA-VI) (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 10 | -10.7 | n/a | n/a | n/a | n/a | n/a | 7.5 | 20 |
Institut des Biomolécules Max Mousseron (IBMM) | Assay Description Inhibition assay using carbonic anhydrases with an SX.18MV-R, a stopped-flow instrument from Applied Photophysics was used for assaying the CA cataly... | Chem Biol Drug Des 74: 636-9 (2009) Article DOI: 10.1111/j.1747-0285.2009.00902.x BindingDB Entry DOI: 10.7270/Q2VX0F1H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase XIII (Mus musculus (mouse)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase XIV (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VB (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VA (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 8.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 5.00E+4 | -5.77 | n/a | n/a | n/a | n/a | n/a | 7.5 | 20 |
Institut des Biomolécules Max Mousseron (IBMM) | Assay Description Inhibition assay using carbonic anhydrases with an SX.18MV-R, a stopped-flow instrument from Applied Photophysics was used for assaying the CA cataly... | Chem Biol Drug Des 74: 636-9 (2009) Article DOI: 10.1111/j.1747-0285.2009.00902.x BindingDB Entry DOI: 10.7270/Q2VX0F1H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM13054 ((2R,4R)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 5.00E+4 | -5.80 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Universite Montpellier II | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 49: 7024-31 (2006) Article DOI: 10.1021/jm060807n BindingDB Entry DOI: 10.7270/Q24T6GKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||