

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM15824 1-{[5-(5-chlorothiophen-2-yl)-1,2-oxazol-3-yl]methyl}-N-(1-ethylpiperidin-4-yl)-1H-indole-2-carboxamide::2-carboxyindole-based inhibitor 8
SMILES: CCN1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(Cl)s1
InChI Key: InChIKey=ZBYUTHMPEVJBEL-UHFFFAOYSA-N
Data: 2 KI
PDB links: 2 PDB IDs contain this monomer as substructures. 2 PDB IDs contain inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coagulation factor X (Homo sapiens (Human)) | BDBM15824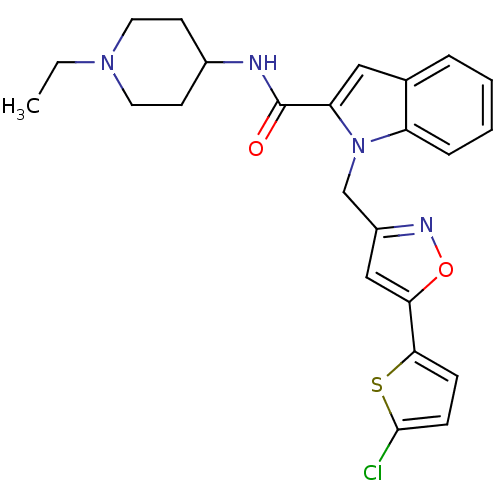 (1-{[5-(5-chlorothiophen-2-yl)-1,2-oxazol-3-yl]meth...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 18 | -10.6 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
Aventis Pharma Deutschland GmbH | Assay Description The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s... | Bioorg Med Chem Lett 14: 4197-201 (2004) Article DOI: 10.1016/j.bmcl.2004.06.019 BindingDB Entry DOI: 10.7270/Q2610XKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM15824 (1-{[5-(5-chlorothiophen-2-yl)-1,2-oxazol-3-yl]meth...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-6.82 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
Aventis Pharma Deutschland GmbH | Assay Description The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge... | Bioorg Med Chem Lett 14: 4197-201 (2004) Article DOI: 10.1016/j.bmcl.2004.06.019 BindingDB Entry DOI: 10.7270/Q2610XKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||