

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM20878 (2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methoxyphenyl)methyl]phenoxy}oxane-3,4,5-triol::CHEMBL270766::O-glucoside, 4a
SMILES: COc1ccc(Cc2ccccc2O[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)cc1
InChI Key: InChIKey=HFLCZNNDZKKXCS-OUUBHVDSSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM20878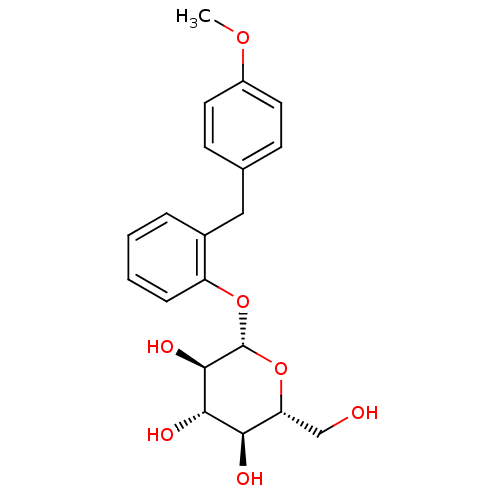 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co Curated by ChEMBL | Assay Description Inhibition of human kidney SGLT2 assessed as renal glucose reabsorption | J Med Chem 52: 1785-94 (2009) Article DOI: 10.1021/jm8013019 BindingDB Entry DOI: 10.7270/Q2F47Q1T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co Curated by ChEMBL | Assay Description Inhibition of human kidney SGLT2 assessed as renal glucose reabsorption | J Med Chem 52: 1785-94 (2009) Article DOI: 10.1021/jm8013019 BindingDB Entry DOI: 10.7270/Q2F47Q1T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 1 (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >8.00E+3 | n/a | n/a | n/a | n/a |
Terns Pharmaceuticals Curated by ChEMBL | Assay Description Inhibition of SGLT1 (unknown origin) | J Med Chem 63: 5031-5073 (2020) Article DOI: 10.1021/acs.jmedchem.9b01701 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Low affinity sodium-glucose cotransporter (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Chugai Pharmaceutical Co., Ltd Curated by ChEMBL | Assay Description Inhibition of human SGLT2 expressed in CHO cells assessed as [14C]AMG uptake after 45 mins | Bioorg Med Chem 19: 5334-41 (2011) Article DOI: 10.1016/j.bmc.2011.08.005 BindingDB Entry DOI: 10.7270/Q2F1903R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 1 (SGLT1) (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chugai Pharmaceutical Co., Ltd Curated by ChEMBL | Assay Description Inhibition of human SGLT1 expressed in CHO cells assessed as [14C]AMG uptake after 45 mins | Bioorg Med Chem 19: 5334-41 (2011) Article DOI: 10.1016/j.bmc.2011.08.005 BindingDB Entry DOI: 10.7270/Q2F1903R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 9.20 | n/a | n/a | n/a | n/a |
Terns Pharmaceuticals Curated by ChEMBL | Assay Description Inhibition of SGLT2 (unknown origin) | J Med Chem 63: 5031-5073 (2020) Article DOI: 10.1021/acs.jmedchem.9b01701 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 9.20 | n/a | n/a | 7.2 | 22 |
Bristol-Myers Squibb Company | Assay Description Inhibitors were assayed for the ability to inhibit [14C]AMG uptake in a protein-free buffer over a 2 h incubation period. The response curve was fitt... | J Med Chem 51: 1145-9 (2008) Article DOI: 10.1021/jm701272q BindingDB Entry DOI: 10.7270/Q2PN93X4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Chugai Pharmaceutical Co. Ltd Curated by ChEMBL | Assay Description Inhibition of human SGLT2 expressed in CHO-K1 cells by [14C]AMG uptake assay | Bioorg Med Chem 20: 4117-27 (2012) Article DOI: 10.1016/j.bmc.2012.04.053 BindingDB Entry DOI: 10.7270/Q26111CJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 1 (SGLT1) (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chugai Pharmaceutical Co. Ltd Curated by ChEMBL | Assay Description Inhibition of human SGLT1 expressed in CHO-K1 cells by [14C]AMG uptake assay | Bioorg Med Chem 20: 4117-27 (2012) Article DOI: 10.1016/j.bmc.2012.04.053 BindingDB Entry DOI: 10.7270/Q26111CJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 1 (SGLT1) (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >8.00E+3 | n/a | n/a | 7.2 | 22 |
Bristol-Myers Squibb Company | Assay Description Inhibitors were assayed for the ability to inhibit [14C]AMG uptake in a protein-free buffer over a 2 h incubation period. The response curve was fitt... | J Med Chem 51: 1145-9 (2008) Article DOI: 10.1021/jm701272q BindingDB Entry DOI: 10.7270/Q2PN93X4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM20878 ((2R,3S,4S,5R,6S)-2-(hydroxymethyl)-6-{2-[(4-methox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.25 | n/a | n/a | n/a | n/a | n/a | n/a |
Green Cross Corporation Curated by ChEMBL | Assay Description Inhibition of human recombinant SGLT2 expressed in CHO cells by liquid scintillation counting | Bioorg Med Chem 18: 2178-94 (2010) Article DOI: 10.1016/j.bmc.2010.01.073 BindingDB Entry DOI: 10.7270/Q2MG7QF6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||