

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50012660 CHEMBL3261078::US9273042, 5::US9556135, 19
SMILES: CC1(C)C(N)=N[C@@](C)(CS1(=O)=O)c1cc(NC(=O)c2ccc(Cl)cn2)ccc1F
InChI Key: InChIKey=LMHDJRUKZXENER-IBGZPJMESA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50012660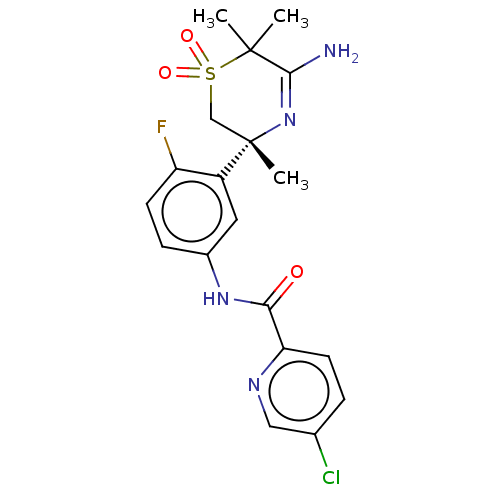 (CHEMBL3261078 | US9273042, 5 | US9556135, 19) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.000900 | n/a | n/a | n/a | n/a | n/a | 37 |
HOFFMANN-LA ROCHE INC. US Patent | Assay Description The Abeta 40 AlphaLISA Assay can be used. The HEK293 APP cells were seeded in 96 well Microtiter plates in cell culture medium (Iscove's, plus 10% (v... | US Patent US9273042 (2016) BindingDB Entry DOI: 10.7270/Q22Z14CV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM50012660 (CHEMBL3261078 | US9273042, 5 | US9556135, 19) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 2.40E+5 | n/a | n/a | n/a | n/a | 3.5 | n/a |
Amgen, Inc. US Patent | Assay Description Recombinant Cat D was expressed in CHO cells. The assay buffer for CathepsinD is 0.05 M citrate pH 3.5, 10% DMSO final, 5 mM CHAPS. The Cat D enzyme ... | US Patent US9556135 (2017) BindingDB Entry DOI: 10.7270/Q2X0691F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50012660 (CHEMBL3261078 | US9273042, 5 | US9556135, 19) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.850 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc. US Patent | Assay Description Several animal models, including mouse, rat, dog, and monkey, may be used to screen for inhibition of beta-secretase activity in vivo following admin... | US Patent US9556135 (2017) BindingDB Entry DOI: 10.7270/Q2X0691F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50012660 (CHEMBL3261078 | US9273042, 5 | US9556135, 19) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development Curated by ChEMBL | Assay Description Inhibition of recombinant soluble human BACE1 catalytic domain using QSY7-EISEVNLDAEFC-Europium-amide as substrate preincubated for 30 mins followed ... | Bioorg Med Chem Lett 24: 2033-45 (2014) Article DOI: 10.1016/j.bmcl.2014.03.025 BindingDB Entry DOI: 10.7270/Q2H41T0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||