

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50102750 CHEMBL3359091
SMILES: COc1cccc(CNC(=O)c2nc3ccc(F)c(OCCc4ccc(cc4)C(O)=O)c3c(=O)[nH]2)c1
InChI Key: InChIKey=YNHGVYGMFAREOF-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Disintegrin and metalloproteinase domain-containing protein 17 (Homo sapiens (Human)) | BDBM50102750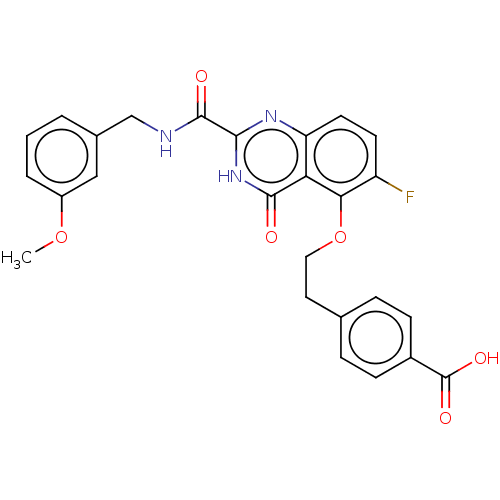 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant TACE using Cy3-PLAQAV(Cy5Q-L-2,3-diaminopropionic acid)-RSSSRNH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-7 (MMP7) (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP7 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase 10 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP10 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP9 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 720 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP8 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-7 (MMP7) (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP7 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Stromelysin-1 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP3 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP2 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP1 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 0.00390 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant MMP13 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP9 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase 10 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP10 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 0.00390 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP13 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Disintegrin and metalloproteinase domain-containing protein 17 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human TACE using Cy3-PLAQAV(Cy5Q-L-2,3-diaminopropionic acid)-RSSSR-NH2 as substrate measured after 40 mins by spectrofluor... | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 720 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP8 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Stromelysin-1 (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP3 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP1 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (MMP14) (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human GST-tagged MMP14 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of recombinant human AMPA-activated MMP2 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate measured after 40 mins by spectrofluorimetric method | Bioorg Med Chem 24: 6149-6165 (2016) Article DOI: 10.1016/j.bmc.2016.09.009 BindingDB Entry DOI: 10.7270/Q2H1341V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (MMP14) (Homo sapiens (Human)) | BDBM50102750 (CHEMBL3359091) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited Curated by ChEMBL | Assay Description Inhibition of human recombinant GST-tagged MMP14 using Cy3-PLGLK(Cy5Q)AR-NH2 as substrate after 40 mins by spectrofluorimetry | J Med Chem 57: 8886-902 (2014) Article DOI: 10.1021/jm500981k BindingDB Entry DOI: 10.7270/Q28P628P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||