 Found 15 hits for monomerid = 50110015
Found 15 hits for monomerid = 50110015 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Urokinase plasminogen activator surface receptor
(Rattus norvegicus) | BDBM50110015
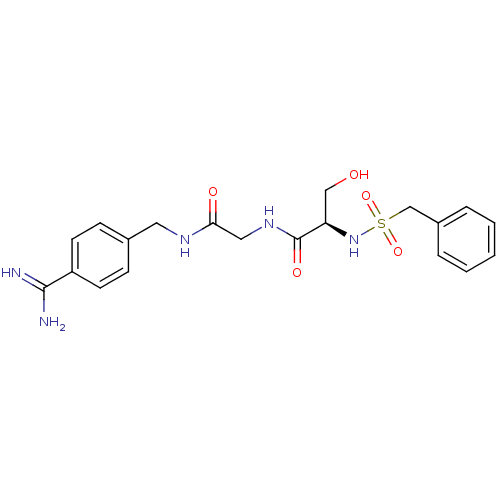
(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 36 | -10.1 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Plasminogen activator urokinase |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Urokinase plasminogen activator surface receptor
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 120 | -9.40 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Plasminogen activator urokinase |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 290 | -8.88 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Trypsin |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 150 | -9.27 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Trypsin |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Prothrombin
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 4.20E+4 | -5.95 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Thrombin |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Prothrombin
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 1.30E+4 | -6.64 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Thrombin |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Plasminogen
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 2.80E+4 | -6.19 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Plasmin |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Plasminogen
(Rattus norvegicus) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 1.10E+4 | -6.74 | n/a | n/a | n/a | n/a | n/a | n/a | 24 |
The Medicines Company
US Patent
| Assay Description
Inhibition constant of the compound against Plasmin |
US Patent US8476306 (2013)
BindingDB Entry DOI: 10.7270/Q2154FPH |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Philipps University Marburg
Curated by ChEMBL
| Assay Description
Inhibition of urokinase-type plasminogen activator (unknown origin) |
J Med Chem 63: 1445-1472 (2020)
Article DOI: 10.1021/acs.jmedchem.9b01060
BindingDB Entry DOI: 10.7270/Q2VQ361P |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine protease 1
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Jena
Curated by ChEMBL
| Assay Description
In vitro inhibition of trypsin. |
Bioorg Med Chem Lett 12: 645-8 (2002)
BindingDB Entry DOI: 10.7270/Q2NZ86XS |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Jena
Curated by ChEMBL
| Assay Description
In vitro inhibition of plasminogen activator urokinase. |
Bioorg Med Chem Lett 12: 645-8 (2002)
BindingDB Entry DOI: 10.7270/Q2NZ86XS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Plasminogen
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Jena
Curated by ChEMBL
| Assay Description
In vitro inhibition of Plasmin. |
Bioorg Med Chem Lett 12: 645-8 (2002)
BindingDB Entry DOI: 10.7270/Q2NZ86XS |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Jena
Curated by ChEMBL
| Assay Description
In vitro inhibition of thrombin. |
Bioorg Med Chem Lett 12: 645-8 (2002)
BindingDB Entry DOI: 10.7270/Q2NZ86XS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Jena
Curated by ChEMBL
| Assay Description
In vitro inhibition of Coagulation factor Xa. |
Bioorg Med Chem Lett 12: 645-8 (2002)
BindingDB Entry DOI: 10.7270/Q2NZ86XS |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50110015

(CHEMBL158936 | N-(BENZYLSULFONYL)SERYL-N~1~-{4-[AM...)Show SMILES NC(=N)c1ccc(CNC(=O)CNC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C20H25N5O5S/c21-19(22)16-8-6-14(7-9-16)10-23-18(27)11-24-20(28)17(12-26)25-31(29,30)13-15-4-2-1-3-5-15/h1-9,17,25-26H,10-13H2,(H3,21,22)(H,23,27)(H,24,28)/t17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Philipps University Marburg
Curated by ChEMBL
| Assay Description
Inhibition of plasmin (unknown origin) |
J Med Chem 63: 1445-1472 (2020)
Article DOI: 10.1021/acs.jmedchem.9b01060
BindingDB Entry DOI: 10.7270/Q2VQ361P |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data