

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50359395 CHEMBL1929422
SMILES: CN(CC#C)Cc1cc2cc(OCCCCC3CCN(Cc4ccccc4)CC3)ccc2n1C
InChI Key: InChIKey=FGYIUWXDEDSRJR-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Monoamine Oxidase Type B (MAO-B) (Rattus norvegicus (rat)) | BDBM50359395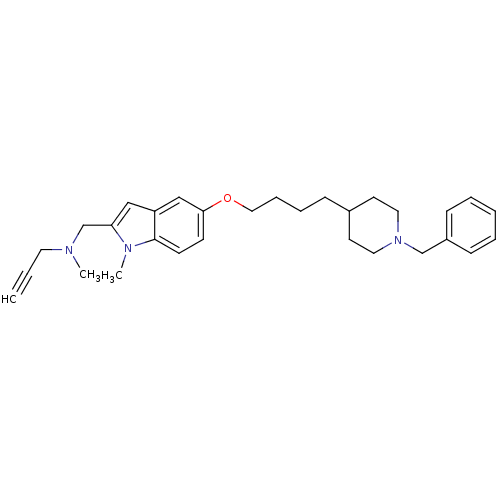 (CHEMBL1929422) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Aut£noma de Barcelona Curated by ChEMBL | Assay Description Inhibition of rat liver mitochondrial MAO-B using [14C]-phenylethylamine after 30 mins by scintillation counting | J Med Chem 54: 8251-70 (2011) Article DOI: 10.1021/jm200853t BindingDB Entry DOI: 10.7270/Q20P10G5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50359395 (CHEMBL1929422) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Aut£noma de Barcelona Curated by ChEMBL | Assay Description Inhibition of Electrophorus electricus AchE using acetylthiocholine iodide as substrate preincubated for 10 mins measured after 15 mins of substrate ... | J Med Chem 54: 8251-70 (2011) Article DOI: 10.1021/jm200853t BindingDB Entry DOI: 10.7270/Q20P10G5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Butyrylcholinesterase (BuChE) (Equus caballus (Horse)) | BDBM50359395 (CHEMBL1929422) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 990 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Aut£noma de Barcelona Curated by ChEMBL | Assay Description Inhibition of equine serum BuchE using butyrylthiocholine iodide as substrate preincubated for 10 mins measured after 15 mins of substrate addition b... | J Med Chem 54: 8251-70 (2011) Article DOI: 10.1021/jm200853t BindingDB Entry DOI: 10.7270/Q20P10G5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoamine oxidase (Rattus norvegicus (rat)) | BDBM50359395 (CHEMBL1929422) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat Aut£noma de Barcelona Curated by ChEMBL | Assay Description Inhibition of rat liver mitochondrial MAO-A using [14C]-5-hydroxytryptamine after 30 mins by scintillation counting | J Med Chem 54: 8251-70 (2011) Article DOI: 10.1021/jm200853t BindingDB Entry DOI: 10.7270/Q20P10G5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||