 Found 95 hits with Last Name = 'narayan' and Initial = 'c'
Found 95 hits with Last Name = 'narayan' and Initial = 'c' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM92964
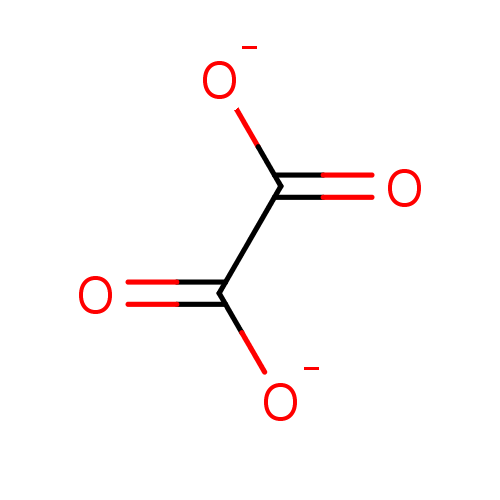
(CHEMBL182928 | Oxalate)Show InChI InChI=1S/C2H2O4/c3-1(4)2(5)6/h(H,3,4)(H,5,6)/p-2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 4.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM92965

(3,3-Difluoroxaloacetate)Show InChI InChI=1S/C4H4F2O6/c5-3(6,1(7)8)4(11,12)2(9)10/h11-12H,(H,7,8)(H,9,10)/p-2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM92966
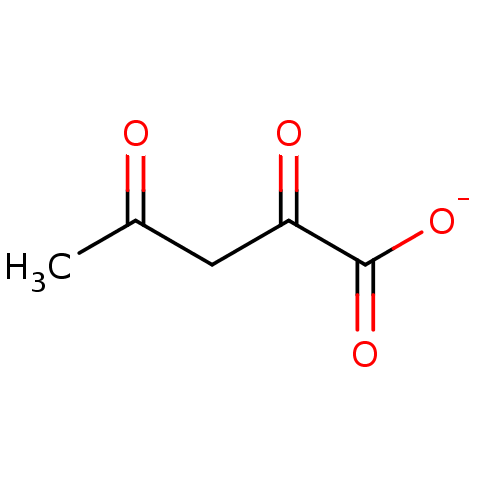
(Acetopyruvate)Show InChI InChI=1S/C5H6O4/c1-3(6)2-4(7)5(8)9/h2H2,1H3,(H,8,9)/p-1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM92966

(Acetopyruvate)Show InChI InChI=1S/C5H6O4/c1-3(6)2-4(7)5(8)9/h2H2,1H3,(H,8,9)/p-1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.09E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM92967
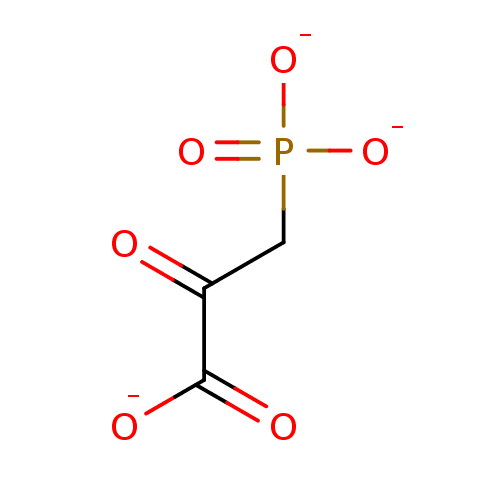
(Phosphonopyruvate)Show InChI InChI=1S/C3H5O6P/c4-2(3(5)6)1-10(7,8)9/h1H2,(H,5,6)(H2,7,8,9)/p-3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 3.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM50273976

(2-oxopentanoate | 2-oxovalerate | alpha-ketovalera...)Show InChI InChI=1S/C5H8O3/c1-2-3-4(6)5(7)8/h2-3H2,1H3,(H,7,8)/p-1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.20E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM50273976

(2-oxopentanoate | 2-oxovalerate | alpha-ketovalera...)Show InChI InChI=1S/C5H8O3/c1-2-3-4(6)5(7)8/h2-3H2,1H3,(H,7,8)/p-1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Oxaloacetate decarboxylase
(Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 /...) | BDBM50159792
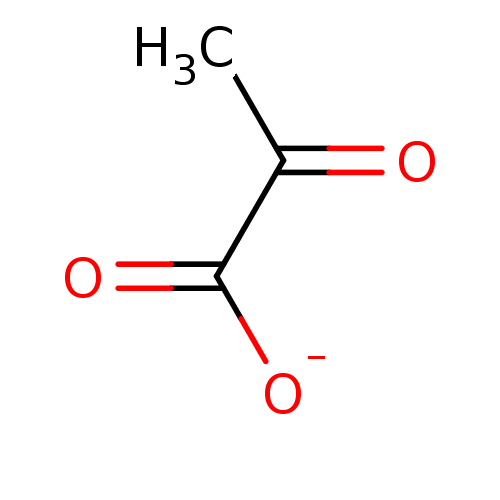
(CHEMBL181886 | Natriumpyruvat | Pyruvate | pyruvic...)Show InChI InChI=1S/C3H4O3/c1-2(4)3(5)6/h1H3,(H,5,6)/p-1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7.20E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland Biotechnology Institute
| Assay Description
Competitive inhibition constant determined for PA4872 catalyzed oxaloacetate decarboxylation. |
Biochemistry 47: 167-82 (2008)
Article DOI: 10.1021/bi701954p
BindingDB Entry DOI: 10.7270/Q20V8BDS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50020412

(CHEMBL3289811)Show SMILES Nc1nc2ccc(OC3CCOC3)nc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F Show InChI InChI=1S/C19H18ClF3N4O3/c20-13-7-10(19(21,22)23)1-2-12(13)15(28)8-27-17-14(25-18(27)24)3-4-16(26-17)30-11-5-6-29-9-11/h1-4,7,11,15,28H,5-6,8-9H2,(H2,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 780 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50020412

(CHEMBL3289811)Show SMILES Nc1nc2ccc(OC3CCOC3)nc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F Show InChI InChI=1S/C19H18ClF3N4O3/c20-13-7-10(19(21,22)23)1-2-12(13)15(28)8-27-17-14(25-18(27)24)3-4-16(26-17)30-11-5-6-29-9-11/h1-4,7,11,15,28H,5-6,8-9H2,(H2,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018442

(CHEMBL3286436)Show SMILES COc1cc(C#N)c2ccc(=O)n(CCN3CCC(CC3)NCc3cnc(C)c(c3)C#N)c2c1 Show InChI InChI=1S/C26H28N6O2/c1-18-20(14-27)11-19(16-29-18)17-30-22-5-7-31(8-6-22)9-10-32-25-13-23(34-2)12-21(15-28)24(25)3-4-26(32)33/h3-4,11-13,16,22,30H,5-10,17H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50020404

(CHEMBL3289803)Show SMILES Nc1nc2ccc(cc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12ClF6N3O/c18-11-5-8(16(19,20)21)1-3-10(11)14(28)7-27-13-6-9(17(22,23)24)2-4-12(13)26-15(27)25/h1-6,14,28H,7H2,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020413
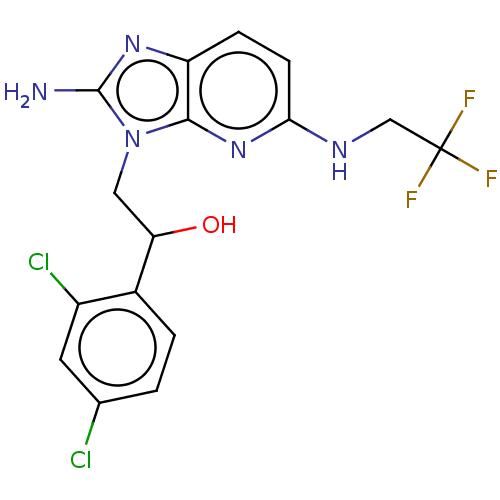
(CHEMBL3289807)Show SMILES Nc1nc2ccc(NCC(F)(F)F)nc2n1CC(O)c1ccc(Cl)cc1Cl Show InChI InChI=1S/C16H14Cl2F3N5O/c17-8-1-2-9(10(18)5-8)12(27)6-26-14-11(24-15(26)22)3-4-13(25-14)23-7-16(19,20)21/h1-5,12,27H,6-7H2,(H2,22,24)(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020414

(CHEMBL3289797)Show InChI InChI=1S/C14H15Cl2N3O/c15-9-3-4-10(11(16)5-9)13(20)7-19-6-12(8-1-2-8)18-14(19)17/h3-6,8,13,20H,1-2,7H2,(H2,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50020411

(CHEMBL3289813)Show SMILES CC(Oc1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1)C(F)(F)F Show InChI InChI=1S/C18H15ClF6N4O2/c1-8(17(20,21)22)31-14-5-4-12-15(28-14)29(16(26)27-12)7-13(30)10-3-2-9(6-11(10)19)18(23,24)25/h2-6,8,13,30H,7H2,1H3,(H2,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50020415
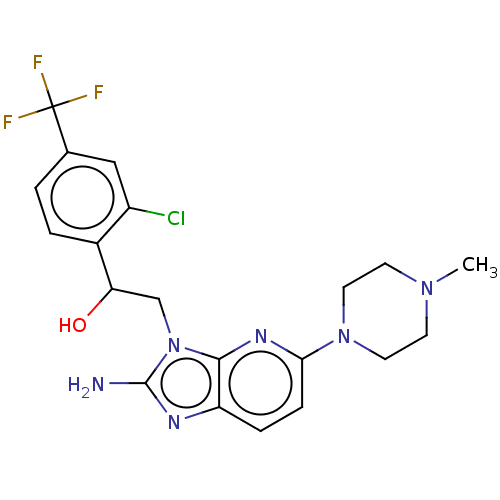
(CHEMBL3289806)Show SMILES CN1CCN(CC1)c1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1 Show InChI InChI=1S/C20H22ClF3N6O/c1-28-6-8-29(9-7-28)17-5-4-15-18(27-17)30(19(25)26-15)11-16(31)13-3-2-12(10-14(13)21)20(22,23)24/h2-5,10,16,31H,6-9,11H2,1H3,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50020408

(CHEMBL3289805)Show InChI InChI=1S/C14H12Cl2N4O/c15-9-4-1-3-8(12(9)16)11(21)7-20-13-10(19-14(20)17)5-2-6-18-13/h1-6,11,21H,7H2,(H2,17,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018290

(CHEMBL3290338)Show SMILES Cc1nc(CNC2CCN(CCn3c4cc(ccc4ccc3=O)C#N)CC2)ccc1C#N Show InChI InChI=1S/C25H26N6O/c1-18-21(16-27)4-6-23(29-18)17-28-22-8-10-30(11-9-22)12-13-31-24-14-19(15-26)2-3-20(24)5-7-25(31)32/h2-7,14,22,28H,8-13,17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50019515

(CHEMBL3291062)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)Nc1cc(=O)n(C)c3cc(F)c(F)cc13)N2C(=O)c1noc(C)c1C |r,TLB:25:24:6.7.8:2.3,9:7:24:2.3| Show InChI InChI=1S/C23H24F2N4O3/c1-11-12(2)32-27-22(11)23(31)29-14-4-5-15(29)7-13(6-14)26-19-10-21(30)28(3)20-9-18(25)17(24)8-16(19)20/h8-10,13-15,26H,4-7H2,1-3H3/t13-,14+,15- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of adenosine A1 receptor (unknown origin) |
J Med Chem 57: 5419-34 (2014)
Article DOI: 10.1021/jm5005978
BindingDB Entry DOI: 10.7270/Q25Q4XNM |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50019516

(CHEMBL3291063)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)Nc1n[nH]c3ccc(F)cc13)N2C(=O)c1cnn(C)c1Cl |r,TLB:21:20:2.3:7.6.8,9:7:20:2.3| Show InChI InChI=1S/C19H20ClFN6O/c1-26-17(20)15(9-22-26)19(28)27-12-3-4-13(27)8-11(7-12)23-18-14-6-10(21)2-5-16(14)24-25-18/h2,5-6,9,11-13H,3-4,7-8H2,1H3,(H2,23,24,25)/t11-,12+,13- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of adenosine A1 receptor (unknown origin) |
J Med Chem 57: 5419-34 (2014)
Article DOI: 10.1021/jm5005978
BindingDB Entry DOI: 10.7270/Q25Q4XNM |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50019519
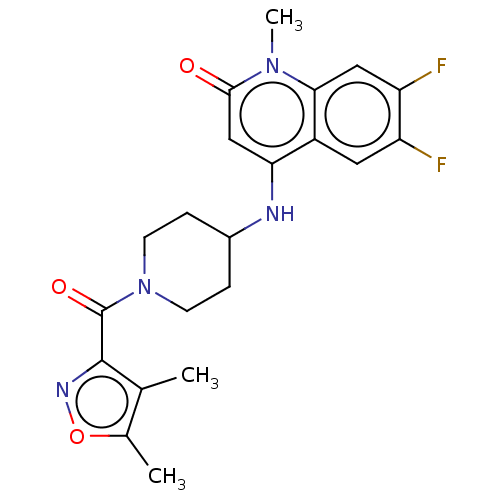
(CHEMBL3291056)Show SMILES Cc1onc(C(=O)N2CCC(CC2)Nc2cc(=O)n(C)c3cc(F)c(F)cc23)c1C Show InChI InChI=1S/C21H22F2N4O3/c1-11-12(2)30-25-20(11)21(29)27-6-4-13(5-7-27)24-17-10-19(28)26(3)18-9-16(23)15(22)8-14(17)18/h8-10,13,24H,4-7H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of adenosine A1 receptor (unknown origin) |
J Med Chem 57: 5419-34 (2014)
Article DOI: 10.1021/jm5005978
BindingDB Entry DOI: 10.7270/Q25Q4XNM |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50019514

(CHEMBL3290772)Show SMILES Cn1ncc(C(=O)N2CCC(CC2)Nc2cc(=O)n(C)c3ccccc23)c1Cl Show InChI InChI=1S/C20H22ClN5O2/c1-24-17-6-4-3-5-14(17)16(11-18(24)27)23-13-7-9-26(10-8-13)20(28)15-12-22-25(2)19(15)21/h3-6,11-13,23H,7-10H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of adenosine A1 receptor (unknown origin) |
J Med Chem 57: 5419-34 (2014)
Article DOI: 10.1021/jm5005978
BindingDB Entry DOI: 10.7270/Q25Q4XNM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018440

(CHEMBL3290342)Show SMILES COc1ccc2n(CCN3CCC(CC3)NCc3cnc(C)c(c3)C#N)c(=O)ccc2n1 Show InChI InChI=1S/C24H28N6O2/c1-17-19(14-25)13-18(15-26-17)16-27-20-7-9-29(10-8-20)11-12-30-22-4-5-23(32-2)28-21(22)3-6-24(30)31/h3-6,13,15,20,27H,7-12,16H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50020408

(CHEMBL3289805)Show InChI InChI=1S/C14H12Cl2N4O/c15-9-4-1-3-8(12(9)16)11(21)7-20-13-10(19-14(20)17)5-2-6-18-13/h1-6,11,21H,7H2,(H2,17,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020416
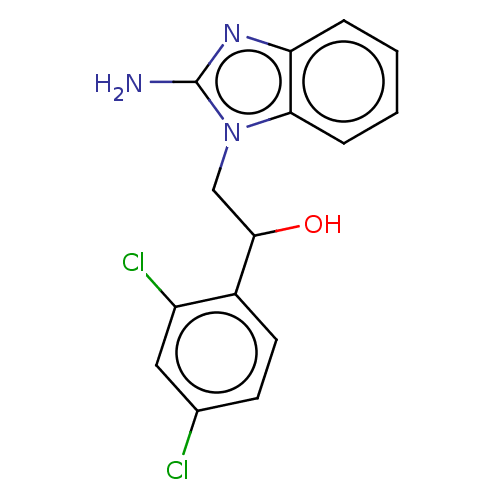
(CHEMBL3289799)Show InChI InChI=1S/C15H13Cl2N3O/c16-9-5-6-10(11(17)7-9)14(21)8-20-13-4-2-1-3-12(13)19-15(20)18/h1-7,14,21H,8H2,(H2,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.03E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020404

(CHEMBL3289803)Show SMILES Nc1nc2ccc(cc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12ClF6N3O/c18-11-5-8(16(19,20)21)1-3-10(11)14(28)7-27-13-6-9(17(22,23)24)2-4-12(13)26-15(27)25/h1-6,14,28H,7H2,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >1.05E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018444
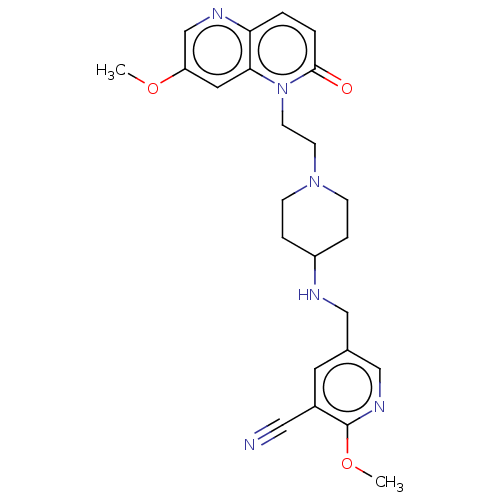
(CHEMBL3290345)Show SMILES COc1cnc2ccc(=O)n(CCN3CCC(CC3)NCc3cnc(OC)c(c3)C#N)c2c1 Show InChI InChI=1S/C24H28N6O3/c1-32-20-12-22-21(27-16-20)3-4-23(31)30(22)10-9-29-7-5-19(6-8-29)26-14-17-11-18(13-25)24(33-2)28-15-17/h3-4,11-12,15-16,19,26H,5-10,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020415

(CHEMBL3289806)Show SMILES CN1CCN(CC1)c1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1 Show InChI InChI=1S/C20H22ClF3N6O/c1-28-6-8-29(9-7-28)17-5-4-15-18(27-17)30(19(25)26-15)11-16(31)13-3-2-12(10-14(13)21)20(22,23)24/h2-5,10,16,31H,6-9,11H2,1H3,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.22E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50020412

(CHEMBL3289811)Show SMILES Nc1nc2ccc(OC3CCOC3)nc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F Show InChI InChI=1S/C19H18ClF3N4O3/c20-13-7-10(19(21,22)23)1-2-12(13)15(28)8-27-17-14(25-18(27)24)3-4-16(26-17)30-11-5-6-29-9-11/h1-4,7,11,15,28H,5-6,8-9H2,(H2,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018447

(CHEMBL3290347)Show SMILES COc1ccc2ncc(=O)n(CCN3CCC(CC3)NCc3cnc(C)c(c3)C#N)c2n1 Show InChI InChI=1S/C23H27N7O2/c1-16-18(12-24)11-17(13-25-16)14-26-19-5-7-29(8-6-19)9-10-30-22(31)15-27-20-3-4-21(32-2)28-23(20)30/h3-4,11,13,15,19,26H,5-10,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50020415

(CHEMBL3289806)Show SMILES CN1CCN(CC1)c1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1 Show InChI InChI=1S/C20H22ClF3N6O/c1-28-6-8-29(9-7-28)17-5-4-15-18(27-17)30(19(25)26-15)11-16(31)13-3-2-12(10-14(13)21)20(22,23)24/h2-5,10,16,31H,6-9,11H2,1H3,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.41E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50019515

(CHEMBL3291062)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)Nc1cc(=O)n(C)c3cc(F)c(F)cc13)N2C(=O)c1noc(C)c1C |r,TLB:25:24:6.7.8:2.3,9:7:24:2.3| Show InChI InChI=1S/C23H24F2N4O3/c1-11-12(2)32-27-22(11)23(31)29-14-4-5-15(29)7-13(6-14)26-19-10-21(30)28(3)20-9-18(25)17(24)8-16(19)20/h8-10,13-15,26H,4-7H2,1-3H3/t13-,14+,15- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by medium-throughput electrophysiology |
J Med Chem 57: 5419-34 (2014)
Article DOI: 10.1021/jm5005978
BindingDB Entry DOI: 10.7270/Q25Q4XNM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020412

(CHEMBL3289811)Show SMILES Nc1nc2ccc(OC3CCOC3)nc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F Show InChI InChI=1S/C19H18ClF3N4O3/c20-13-7-10(19(21,22)23)1-2-12(13)15(28)8-27-17-14(25-18(27)24)3-4-16(26-17)30-11-5-6-29-9-11/h1-4,7,11,15,28H,5-6,8-9H2,(H2,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.66E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50020411

(CHEMBL3289813)Show SMILES CC(Oc1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1)C(F)(F)F Show InChI InChI=1S/C18H15ClF6N4O2/c1-8(17(20,21)22)31-14-5-4-12-15(28-14)29(16(26)27-12)7-13(30)10-3-2-9(6-11(10)19)18(23,24)25/h2-6,8,13,30H,7H2,1H3,(H2,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.03E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018288
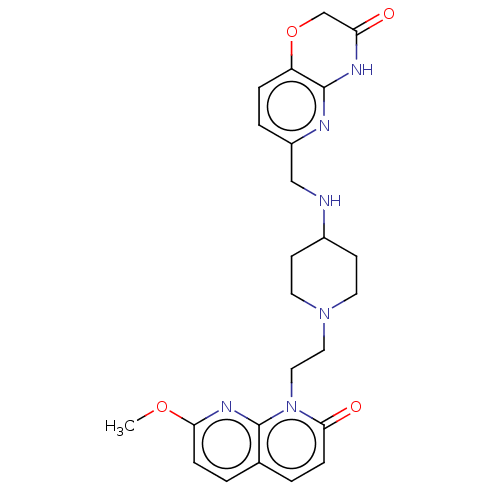
(CHEMBL3290336)Show SMILES COc1ccc2ccc(=O)n(CCN3CCC(CC3)NCc3ccc4OCC(=O)Nc4n3)c2n1 Show InChI InChI=1S/C24H28N6O4/c1-33-21-6-2-16-3-7-22(32)30(24(16)28-21)13-12-29-10-8-17(9-11-29)25-14-18-4-5-19-23(26-18)27-20(31)15-34-19/h2-7,17,25H,8-15H2,1H3,(H,26,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50019511

(CHEMBL3290766)Show SMILES Cn1ncc(C(=O)N2CCC(CC2)Nc2cc(=O)[nH]c3ccccc23)c1Cl Show InChI InChI=1S/C19H20ClN5O2/c1-24-18(20)14(11-21-24)19(27)25-8-6-12(7-9-25)22-16-10-17(26)23-15-5-3-2-4-13(15)16/h2-5,10-12H,6-9H2,1H3,(H2,22,23,26) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.19E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of mu opioid receptor (unknown origin) |
J Med Chem 57: 5419-34 (2014)
Article DOI: 10.1021/jm5005978
BindingDB Entry DOI: 10.7270/Q25Q4XNM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020417

(CHEMBL3289794)Show InChI InChI=1S/C11H11Cl2N3O/c12-8-3-1-2-7(10(8)13)9(17)6-16-5-4-15-11(16)14/h1-5,9,17H,6H2,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50020411

(CHEMBL3289813)Show SMILES CC(Oc1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1)C(F)(F)F Show InChI InChI=1S/C18H15ClF6N4O2/c1-8(17(20,21)22)31-14-5-4-12-15(28-14)29(16(26)27-12)7-13(30)10-3-2-9(6-11(10)19)18(23,24)25/h2-6,8,13,30H,7H2,1H3,(H2,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50020418
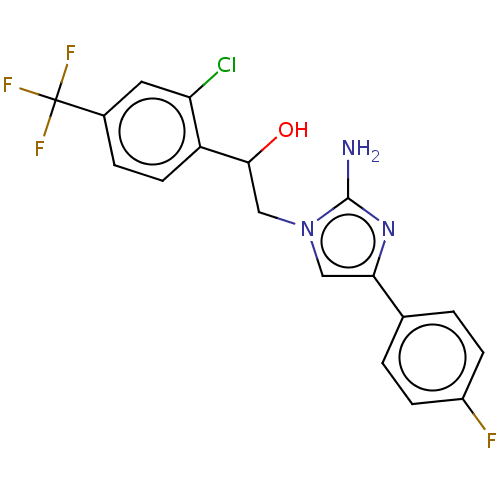
(CHEMBL3289798)Show SMILES Nc1nc(cn1CC(O)c1ccc(cc1Cl)C(F)(F)F)-c1ccc(F)cc1 Show InChI InChI=1S/C18H14ClF4N3O/c19-14-7-11(18(21,22)23)3-6-13(14)16(27)9-26-8-15(25-17(26)24)10-1-4-12(20)5-2-10/h1-8,16,27H,9H2,(H2,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50018407
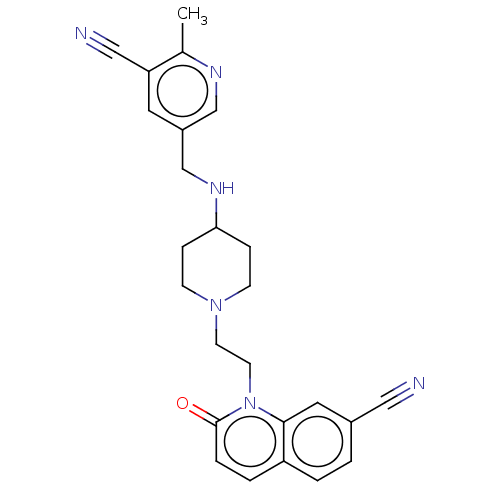
(CHEMBL3290339)Show SMILES Cc1ncc(CNC2CCN(CCn3c4cc(ccc4ccc3=O)C#N)CC2)cc1C#N Show InChI InChI=1S/C25H26N6O/c1-18-22(15-27)12-20(16-28-18)17-29-23-6-8-30(9-7-23)10-11-31-24-13-19(14-26)2-3-21(24)4-5-25(31)32/h2-5,12-13,16,23,29H,6-11,17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp electrophysiological analysis |
J Med Chem 57: 4889-905 (2014)
Article DOI: 10.1021/jm500432n
BindingDB Entry DOI: 10.7270/Q2416ZM4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50020404

(CHEMBL3289803)Show SMILES Nc1nc2ccc(cc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12ClF6N3O/c18-11-5-8(16(19,20)21)1-3-10(11)14(28)7-27-13-6-9(17(22,23)24)2-4-12(13)26-15(27)25/h1-6,14,28H,7H2,(H2,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.63E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50020404

(CHEMBL3289803)Show SMILES Nc1nc2ccc(cc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12ClF6N3O/c18-11-5-8(16(19,20)21)1-3-10(11)14(28)7-27-13-6-9(17(22,23)24)2-4-12(13)26-15(27)25/h1-6,14,28H,7H2,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.79E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50020415

(CHEMBL3289806)Show SMILES CN1CCN(CC1)c1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1 Show InChI InChI=1S/C20H22ClF3N6O/c1-28-6-8-29(9-7-28)17-5-4-15-18(27-17)30(19(25)26-15)11-16(31)13-3-2-12(10-14(13)21)20(22,23)24/h2-5,10,16,31H,6-9,11H2,1H3,(H2,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.79E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50020411

(CHEMBL3289813)Show SMILES CC(Oc1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1)C(F)(F)F Show InChI InChI=1S/C18H15ClF6N4O2/c1-8(17(20,21)22)31-14-5-4-12-15(28-14)29(16(26)27-12)7-13(30)10-3-2-9(6-11(10)19)18(23,24)25/h2-6,8,13,30H,7H2,1H3,(H2,26,27) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.83E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50020404

(CHEMBL3289803)Show SMILES Nc1nc2ccc(cc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H12ClF6N3O/c18-11-5-8(16(19,20)21)1-3-10(11)14(28)7-27-13-6-9(17(22,23)24)2-4-12(13)26-15(27)25/h1-6,14,28H,7H2,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.94E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50020412

(CHEMBL3289811)Show SMILES Nc1nc2ccc(OC3CCOC3)nc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F Show InChI InChI=1S/C19H18ClF3N4O3/c20-13-7-10(19(21,22)23)1-2-12(13)15(28)8-27-17-14(25-18(27)24)3-4-16(26-17)30-11-5-6-29-9-11/h1-4,7,11,15,28H,5-6,8-9H2,(H2,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50020415

(CHEMBL3289806)Show SMILES CN1CCN(CC1)c1ccc2nc(N)n(CC(O)c3ccc(cc3Cl)C(F)(F)F)c2n1 Show InChI InChI=1S/C20H22ClF3N6O/c1-28-6-8-29(9-7-28)17-5-4-15-18(27-17)30(19(25)26-15)11-16(31)13-3-2-12(10-14(13)21)20(22,23)24/h2-5,10,16,31H,6-9,11H2,1H3,(H2,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50020412

(CHEMBL3289811)Show SMILES Nc1nc2ccc(OC3CCOC3)nc2n1CC(O)c1ccc(cc1Cl)C(F)(F)F Show InChI InChI=1S/C19H18ClF3N4O3/c20-13-7-10(19(21,22)23)1-2-12(13)15(28)8-27-17-14(25-18(27)24)3-4-16(26-17)30-11-5-6-29-9-11/h1-4,7,11,15,28H,5-6,8-9H2,(H2,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50020408

(CHEMBL3289805)Show InChI InChI=1S/C14H12Cl2N4O/c15-9-4-1-3-8(12(9)16)11(21)7-20-13-10(19-14(20)17)5-2-6-18-13/h1-6,11,21H,7H2,(H2,17,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50020408

(CHEMBL3289805)Show InChI InChI=1S/C14H12Cl2N4O/c15-9-4-1-3-8(12(9)16)11(21)7-20-13-10(19-14(20)17)5-2-6-18-13/h1-6,11,21H,7H2,(H2,17,19) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 5702-13 (2014)
Article DOI: 10.1021/jm500535j
BindingDB Entry DOI: 10.7270/Q2X3501C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data