 Found 114 hits with Last Name = 'chattopadhyay' and Initial = 'd'
Found 114 hits with Last Name = 'chattopadhyay' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320792
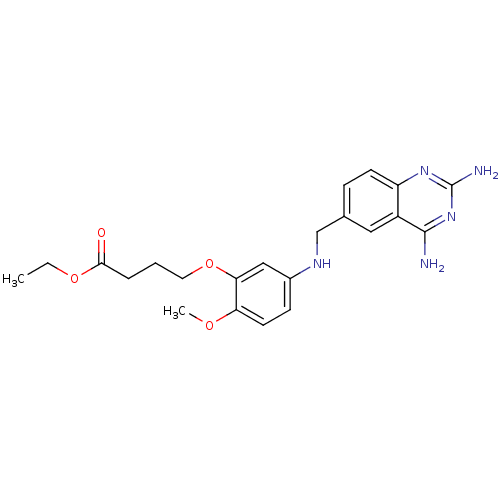
(CHEMBL1165388 | Ethyl4-(5-[(2,4-diamino-6-quinazol...)Show SMILES CCOC(=O)CCCOc1cc(NCc2ccc3nc(N)nc(N)c3c2)ccc1OC Show InChI InChI=1S/C22H27N5O4/c1-3-30-20(28)5-4-10-31-19-12-15(7-9-18(19)29-2)25-13-14-6-8-17-16(11-14)21(23)27-22(24)26-17/h6-9,11-12,25H,3-5,10,13H2,1-2H3,(H4,23,24,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320791
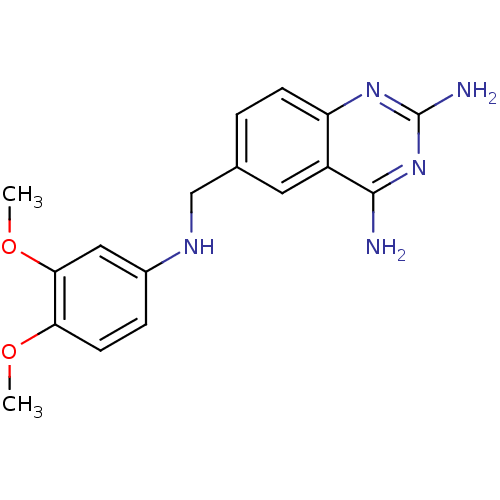
(6-[(3,4-Dimethoxyanilino)methyl]-2,4-quinazolinedi...)Show InChI InChI=1S/C17H19N5O2/c1-23-14-6-4-11(8-15(14)24-2)20-9-10-3-5-13-12(7-10)16(18)22-17(19)21-13/h3-8,20H,9H2,1-2H3,(H4,18,19,21,22) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320793
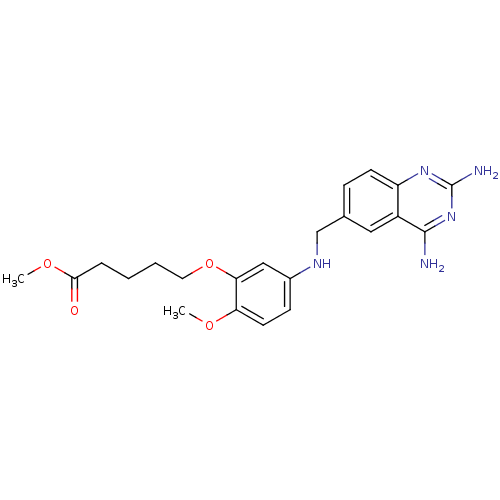
(CHEMBL1165305 | Methyl5-(5-[(2,4-diamino-6-quinazo...)Show SMILES COC(=O)CCCCOc1cc(NCc2ccc3nc(N)nc(N)c3c2)ccc1OC Show InChI InChI=1S/C22H27N5O4/c1-29-18-9-7-15(12-19(18)31-10-4-3-5-20(28)30-2)25-13-14-6-8-17-16(11-14)21(23)27-22(24)26-17/h6-9,11-12,25H,3-5,10,13H2,1-2H3,(H4,23,24,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131975

(1-(2-Methylamino-3-phenyl-propionyl)-pyrrolidine-2...)Show SMILES CN[C@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C28H35N7O3S/c1-31-21(17-18-9-3-2-4-10-18)27(38)35-16-8-13-22(35)25(37)33-20(12-7-15-32-28(29)30)24(36)26-34-19-11-5-6-14-23(19)39-26/h2-6,9-11,14,20-22,31H,7-8,12-13,15-17H2,1H3,(H,33,37)(H4,29,30,32)/t20-,21+,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320794
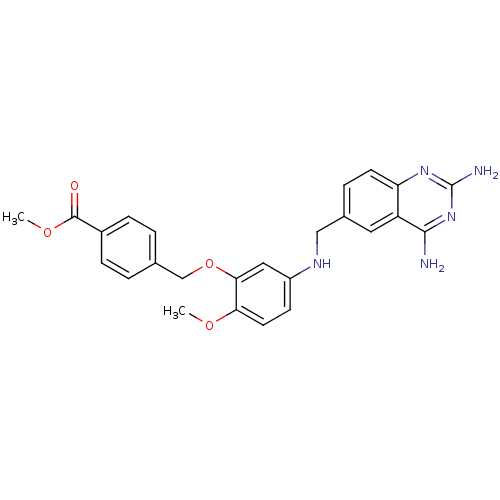
(CHEMBL1165306 | Methyl4-[(5-[(2,4-diamino-6-quinaz...)Show SMILES COC(=O)c1ccc(COc2cc(NCc3ccc4nc(N)nc(N)c4c3)ccc2OC)cc1 Show InChI InChI=1S/C25H25N5O4/c1-32-21-10-8-18(12-22(21)34-14-15-3-6-17(7-4-15)24(31)33-2)28-13-16-5-9-20-19(11-16)23(26)30-25(27)29-20/h3-12,28H,13-14H2,1-2H3,(H4,26,27,29,30) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320790
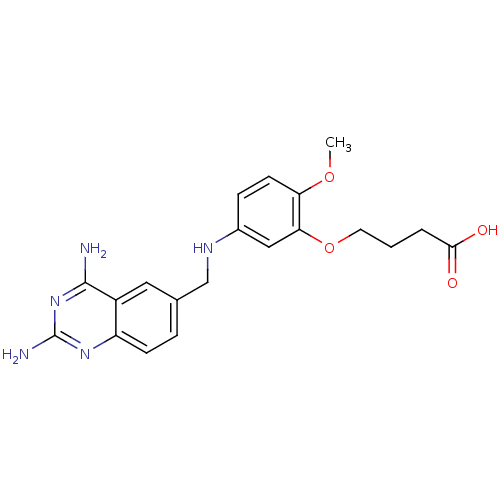
(4-(5-[(2,4-diamino-6-quinazolinyl)methyl]amino-2-m...)Show SMILES COc1ccc(NCc2ccc3nc(N)nc(N)c3c2)cc1OCCCC(O)=O Show InChI InChI=1S/C20H23N5O4/c1-28-16-7-5-13(10-17(16)29-8-2-3-18(26)27)23-11-12-4-6-15-14(9-12)19(21)25-20(22)24-15/h4-7,9-10,23H,2-3,8,11H2,1H3,(H,26,27)(H4,21,22,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131980

(1-Acetyl-pyrrolidine-2-carboxylic acid [1-(benzoth...)Show SMILES CC(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O3S/c1-12(27)26-11-5-8-15(26)18(29)24-14(7-4-10-23-20(21)22)17(28)19-25-13-6-2-3-9-16(13)30-19/h2-3,6,9,14-15H,4-5,7-8,10-11H2,1H3,(H,24,29)(H4,21,22,23)/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320791

(6-[(3,4-Dimethoxyanilino)methyl]-2,4-quinazolinedi...)Show InChI InChI=1S/C17H19N5O2/c1-23-14-6-4-11(8-15(14)24-2)20-9-10-3-5-13-12(7-10)16(18)22-17(19)21-13/h3-8,20H,9H2,1-2H3,(H4,18,19,21,22) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by Lineweaver-Burk plot |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320792

(CHEMBL1165388 | Ethyl4-(5-[(2,4-diamino-6-quinazol...)Show SMILES CCOC(=O)CCCOc1cc(NCc2ccc3nc(N)nc(N)c3c2)ccc1OC Show InChI InChI=1S/C22H27N5O4/c1-3-30-20(28)5-4-10-31-19-12-15(7-9-18(19)29-2)25-13-14-6-8-17-16(11-14)21(23)27-22(24)26-17/h6-9,11-12,25H,3-5,10,13H2,1-2H3,(H4,23,24,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 5.68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by Lineweaver-Burk plot |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131977

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,12,14-15,28H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131975

(1-(2-Methylamino-3-phenyl-propionyl)-pyrrolidine-2...)Show SMILES CN[C@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C28H35N7O3S/c1-31-21(17-18-9-3-2-4-10-18)27(38)35-16-8-13-22(35)25(37)33-20(12-7-15-32-28(29)30)24(36)26-34-19-11-5-6-14-23(19)39-26/h2-6,9-11,14,20-22,31H,7-8,12-13,15-17H2,1H3,(H,33,37)(H4,29,30,32)/t20-,21+,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM18268
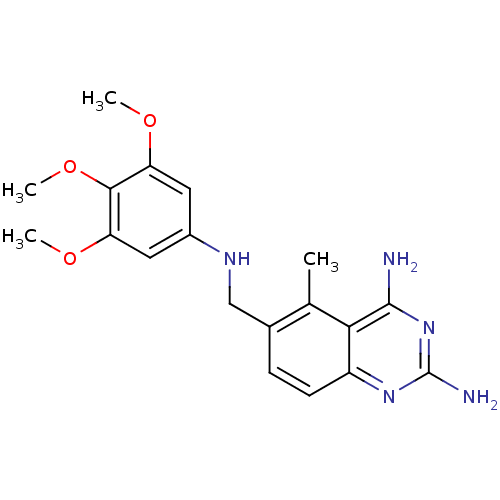
(5-methyl-6-{[(3,4,5-trimethoxyphenyl)amino]methyl}...)Show InChI InChI=1S/C19H23N5O3/c1-10-11(5-6-13-16(10)18(20)24-19(21)23-13)9-22-12-7-14(25-2)17(27-4)15(8-12)26-3/h5-8,22H,9H2,1-4H3,(H4,20,21,23,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 6.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131982
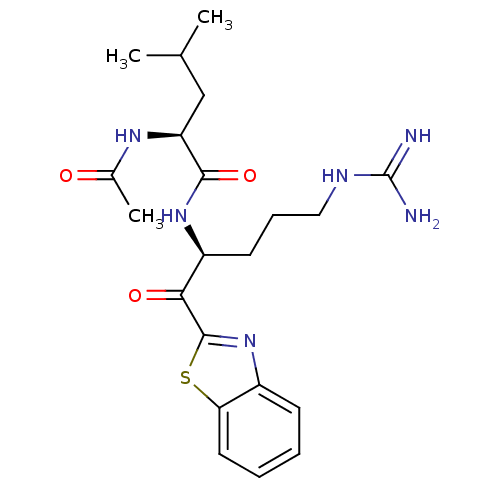
(2-Acetylamino-4-methyl-pentanoic acid [1-(benzothi...)Show SMILES CC(C)C[C@H](NC(C)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C21H30N6O3S/c1-12(2)11-16(25-13(3)28)19(30)26-15(8-6-10-24-21(22)23)18(29)20-27-14-7-4-5-9-17(14)31-20/h4-5,7,9,12,15-16H,6,8,10-11H2,1-3H3,(H,25,28)(H,26,30)(H4,22,23,24)/t15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131989

(1-Acetyl-2,5-dihydro-1H-pyrrole-2-carboxylic acid ...)Show SMILES CC(=O)N1CC=C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 |c:5| Show InChI InChI=1S/C20H24N6O3S/c1-12(27)26-11-5-8-15(26)18(29)24-14(7-4-10-23-20(21)22)17(28)19-25-13-6-2-3-9-16(13)30-19/h2-3,5-6,8-9,14-15H,4,7,10-11H2,1H3,(H,24,29)(H4,21,22,23)/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50320792

(CHEMBL1165388 | Ethyl4-(5-[(2,4-diamino-6-quinazol...)Show SMILES CCOC(=O)CCCOc1cc(NCc2ccc3nc(N)nc(N)c3c2)ccc1OC Show InChI InChI=1S/C22H27N5O4/c1-3-30-20(28)5-4-10-31-19-12-15(7-9-18(19)29-2)25-13-14-6-8-17-16(11-14)21(23)27-22(24)26-17/h6-9,11-12,25H,3-5,10,13H2,1-2H3,(H4,23,24,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 9.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of human DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131983

(1-Acetyl-4-oxo-pyrrolidine-2-carboxylic acid [1-(b...)Show SMILES CC(=O)N1CC(=O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H24N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,14-15H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50320791

(6-[(3,4-Dimethoxyanilino)methyl]-2,4-quinazolinedi...)Show InChI InChI=1S/C17H19N5O2/c1-23-14-6-4-11(8-15(14)24-2)20-9-10-3-5-13-12(7-10)16(18)22-17(19)21-13/h3-8,20H,9H2,1-2H3,(H4,18,19,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of human DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50320793

(CHEMBL1165305 | Methyl5-(5-[(2,4-diamino-6-quinazo...)Show SMILES COC(=O)CCCCOc1cc(NCc2ccc3nc(N)nc(N)c3c2)ccc1OC Show InChI InChI=1S/C22H27N5O4/c1-29-18-9-7-15(12-19(18)31-10-4-3-5-20(28)30-2)25-13-14-6-8-17-16(11-14)21(23)27-22(24)26-17/h6-9,11-12,25H,3-5,10,13H2,1-2H3,(H4,23,24,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of human DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131985
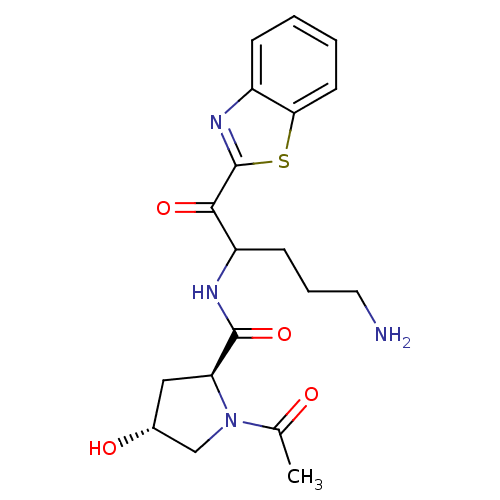
(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)NC(CCCN)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H24N4O4S/c1-11(24)23-10-12(25)9-15(23)18(27)21-14(6-4-8-20)17(26)19-22-13-5-2-3-7-16(13)28-19/h2-3,5,7,12,14-15,25H,4,6,8-10,20H2,1H3,(H,21,27)/t12-,14?,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131980

(1-Acetyl-pyrrolidine-2-carboxylic acid [1-(benzoth...)Show SMILES CC(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O3S/c1-12(27)26-11-5-8-15(26)18(29)24-14(7-4-10-23-20(21)22)17(28)19-25-13-6-2-3-9-16(13)30-19/h2-3,6,9,14-15H,4-5,7-8,10-11H2,1H3,(H,24,29)(H4,21,22,23)/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131984
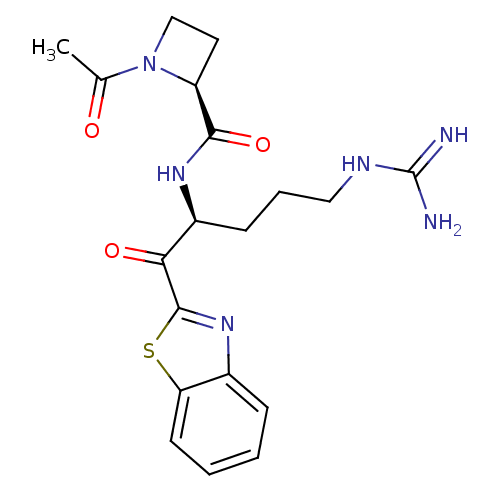
(1-Acetyl-azetidine-2-carboxylic acid [1-(benzothia...)Show SMILES CC(=O)N1CC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H24N6O3S/c1-11(26)25-10-8-14(25)17(28)23-13(6-4-9-22-19(20)21)16(27)18-24-12-5-2-3-7-15(12)29-18/h2-3,5,7,13-14H,4,6,8-10H2,1H3,(H,23,28)(H4,20,21,22)/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131977

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,12,14-15,28H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50320794

(CHEMBL1165306 | Methyl4-[(5-[(2,4-diamino-6-quinaz...)Show SMILES COC(=O)c1ccc(COc2cc(NCc3ccc4nc(N)nc(N)c4c3)ccc2OC)cc1 Show InChI InChI=1S/C25H25N5O4/c1-32-21-10-8-18(12-22(21)34-14-15-3-6-17(7-4-15)24(31)33-2)28-13-16-5-9-20-19(11-16)23(26)30-25(27)29-20/h3-12,28H,13-14H2,1-2H3,(H4,26,27,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 24.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of human DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma cruzi) | BDBM50320789
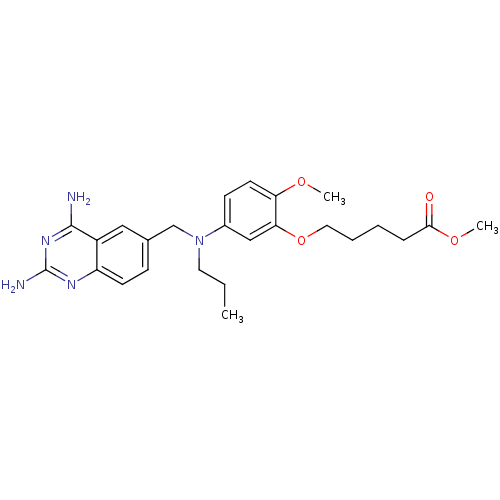
(CHEMBL1164234 | methyl5-{5-[[(2,4-diamino-6-quinaz...)Show SMILES CCCN(Cc1ccc2nc(N)nc(N)c2c1)c1ccc(OC)c(OCCCCC(=O)OC)c1 Show InChI InChI=1S/C25H33N5O4/c1-4-12-30(16-17-8-10-20-19(14-17)24(26)29-25(27)28-20)18-9-11-21(32-2)22(15-18)34-13-6-5-7-23(31)33-3/h8-11,14-15H,4-7,12-13,16H2,1-3H3,(H4,26,27,28,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 24.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma cruzi DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131979

(CHEMBL340547 | Cyclopentanecarboxylic acid [1-(ben...)Show SMILES NC(=N)NCCCC(NC(=O)C1CCCC1)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H25N5O2S/c20-19(21)22-11-5-9-14(23-17(26)12-6-1-2-7-12)16(25)18-24-13-8-3-4-10-15(13)27-18/h3-4,8,10,12,14H,1-2,5-7,9,11H2,(H,23,26)(H4,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131988
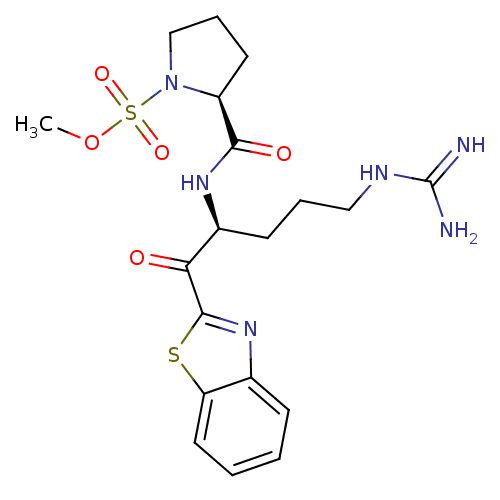
(2-[1-(Benzothiazole-2-carbonyl)-4-guanidino-butylc...)Show SMILES COS(=O)(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H26N6O5S2/c1-30-32(28,29)25-11-5-8-14(25)17(27)23-13(7-4-10-22-19(20)21)16(26)18-24-12-6-2-3-9-15(12)31-18/h2-3,6,9,13-14H,4-5,7-8,10-11H2,1H3,(H,23,27)(H4,20,21,22)/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131988

(2-[1-(Benzothiazole-2-carbonyl)-4-guanidino-butylc...)Show SMILES COS(=O)(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H26N6O5S2/c1-30-32(28,29)25-11-5-8-14(25)17(27)23-13(7-4-10-22-19(20)21)16(26)18-24-12-6-2-3-9-15(12)31-18/h2-3,6,9,13-14H,4-5,7-8,10-11H2,1H3,(H,23,27)(H4,20,21,22)/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131989

(1-Acetyl-2,5-dihydro-1H-pyrrole-2-carboxylic acid ...)Show SMILES CC(=O)N1CC=C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 |c:5| Show InChI InChI=1S/C20H24N6O3S/c1-12(27)26-11-5-8-15(26)18(29)24-14(7-4-10-23-20(21)22)17(28)19-25-13-6-2-3-9-16(13)30-19/h2-3,5-6,8-9,14-15H,4,7,10-11H2,1H3,(H,24,29)(H4,21,22,23)/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131982

(2-Acetylamino-4-methyl-pentanoic acid [1-(benzothi...)Show SMILES CC(C)C[C@H](NC(C)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C21H30N6O3S/c1-12(2)11-16(25-13(3)28)19(30)26-15(8-6-10-24-21(22)23)18(29)20-27-14-7-4-5-9-17(14)31-20/h4-5,7,9,12,15-16H,6,8,10-11H2,1-3H3,(H,25,28)(H,26,30)(H4,22,23,24)/t15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50320790

(4-(5-[(2,4-diamino-6-quinazolinyl)methyl]amino-2-m...)Show SMILES COc1ccc(NCc2ccc3nc(N)nc(N)c3c2)cc1OCCCC(O)=O Show InChI InChI=1S/C20H23N5O4/c1-28-16-7-5-13(10-17(16)29-8-2-3-18(26)27)23-11-12-4-6-15-14(9-12)19(21)25-20(22)24-15/h4-7,9-10,23H,2-3,8,11H2,1H3,(H,26,27)(H4,21,22,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 49.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of human DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131984

(1-Acetyl-azetidine-2-carboxylic acid [1-(benzothia...)Show SMILES CC(=O)N1CC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H24N6O3S/c1-11(26)25-10-8-14(25)17(28)23-13(6-4-9-22-19(20)21)16(27)18-24-12-5-2-3-7-15(12)29-18/h2-3,5,7,13-14H,4,6,8-10H2,1H3,(H,23,28)(H4,20,21,22)/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131983

(1-Acetyl-4-oxo-pyrrolidine-2-carboxylic acid [1-(b...)Show SMILES CC(=O)N1CC(=O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H24N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,14-15H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 73 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131974
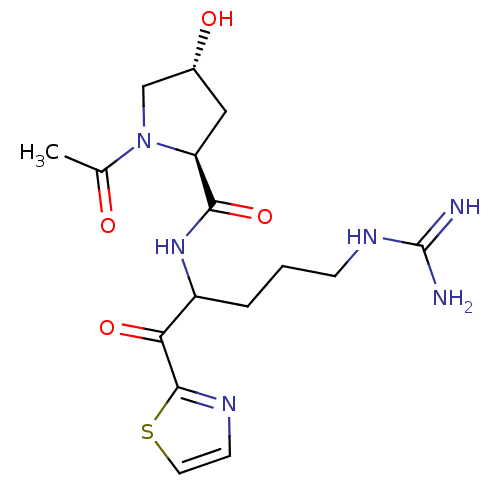
(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)NC(CCCNC(N)=N)C(=O)c1nccs1 Show InChI InChI=1S/C16H24N6O4S/c1-9(23)22-8-10(24)7-12(22)14(26)21-11(3-2-4-20-16(17)18)13(25)15-19-5-6-27-15/h5-6,10-12,24H,2-4,7-8H2,1H3,(H,21,26)(H4,17,18,20)/t10-,11?,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 81 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131979

(CHEMBL340547 | Cyclopentanecarboxylic acid [1-(ben...)Show SMILES NC(=N)NCCCC(NC(=O)C1CCCC1)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H25N5O2S/c20-19(21)22-11-5-9-14(23-17(26)12-6-1-2-7-12)16(25)18-24-13-8-3-4-10-15(13)27-18/h3-4,8,10,12,14H,1-2,5-7,9,11H2,(H,23,26)(H4,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 88 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131985

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)NC(CCCN)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C19H24N4O4S/c1-11(24)23-10-12(25)9-15(23)18(27)21-14(6-4-8-20)17(26)19-22-13-5-2-3-7-16(13)28-19/h2-3,5,7,12,14-15,25H,4,6,8-10,20H2,1H3,(H,21,27)/t12-,14?,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM16127
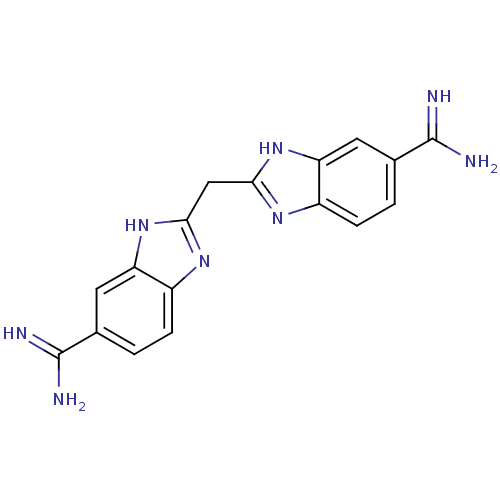
(2,2 -methanediylbis(1H-benzimidazole-6-carboximida...)Show SMILES NC(=N)c1ccc2nc(Cc3nc4ccc(cc4[nH]3)C(N)=N)[nH]c2c1 Show InChI InChI=1S/C17H16N8/c18-16(19)8-1-3-10-12(5-8)24-14(22-10)7-15-23-11-4-2-9(17(20)21)6-13(11)25-15/h1-6H,7H2,(H3,18,19)(H3,20,21)(H,22,24)(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM16127

(2,2 -methanediylbis(1H-benzimidazole-6-carboximida...)Show SMILES NC(=N)c1ccc2nc(Cc3nc4ccc(cc4[nH]3)C(N)=N)[nH]c2c1 Show InChI InChI=1S/C17H16N8/c18-16(19)8-1-3-10-12(5-8)24-14(22-10)7-15-23-11-4-2-9(17(20)21)6-13(11)25-15/h1-6H,7H2,(H3,18,19)(H3,20,21)(H,22,24)(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131978
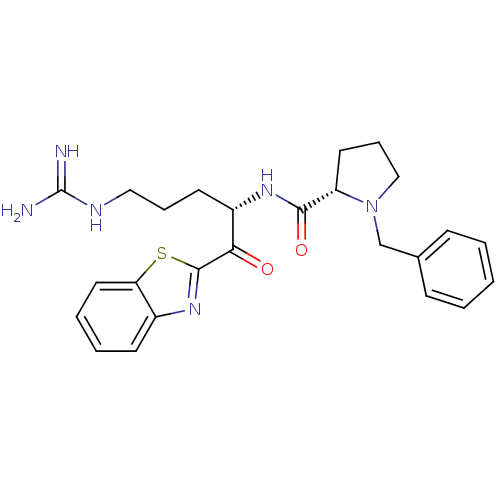
(1-Benzyl-pyrrolidine-2-carboxylic acid [1-(benzoth...)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@@H]1CCCN1Cc1ccccc1)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C25H30N6O2S/c26-25(27)28-14-6-11-19(22(32)24-30-18-10-4-5-13-21(18)34-24)29-23(33)20-12-7-15-31(20)16-17-8-2-1-3-9-17/h1-5,8-10,13,19-20H,6-7,11-12,14-16H2,(H,29,33)(H4,26,27,28)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131977

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,12,14-15,28H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131977

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,12,14-15,28H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131974

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)NC(CCCNC(N)=N)C(=O)c1nccs1 Show InChI InChI=1S/C16H24N6O4S/c1-9(23)22-8-10(24)7-12(22)14(26)21-11(3-2-4-20-16(17)18)13(25)15-19-5-6-27-15/h5-6,10-12,24H,2-4,7-8H2,1H3,(H,21,26)(H4,17,18,20)/t10-,11?,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50320789

(CHEMBL1164234 | methyl5-{5-[[(2,4-diamino-6-quinaz...)Show SMILES CCCN(Cc1ccc2nc(N)nc(N)c2c1)c1ccc(OC)c(OCCCCC(=O)OC)c1 Show InChI InChI=1S/C25H33N5O4/c1-4-12-30(16-17-8-10-20-19(14-17)24(26)29-25(27)28-20)18-9-11-21(32-2)22(15-18)34-13-6-5-7-23(31)33-3/h8-11,14-15H,4-7,12-13,16H2,1-3H3,(H4,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 188 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama
Curated by ChEMBL
| Assay Description
Inhibition of human DHFR by spectrophotometric assay |
Bioorg Med Chem 18: 4056-66 (2010)
Article DOI: 10.1016/j.bmc.2010.04.020
BindingDB Entry DOI: 10.7270/Q26110GG |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131978

(1-Benzyl-pyrrolidine-2-carboxylic acid [1-(benzoth...)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@@H]1CCCN1Cc1ccccc1)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C25H30N6O2S/c26-25(27)28-14-6-11-19(22(32)24-30-18-10-4-5-13-21(18)34-24)29-23(33)20-12-7-15-31(20)16-17-8-2-1-3-9-17/h1-5,8-10,13,19-20H,6-7,11-12,14-16H2,(H,29,33)(H4,26,27,28)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131981
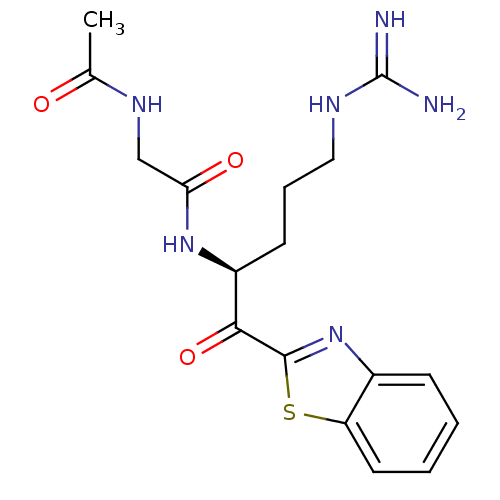
(CHEMBL340098 | N-{[1-(Benzothiazole-2-carbonyl)-4-...)Show SMILES CC(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C17H22N6O3S/c1-10(24)21-9-14(25)22-12(6-4-8-20-17(18)19)15(26)16-23-11-5-2-3-7-13(11)27-16/h2-3,5,7,12H,4,6,8-9H2,1H3,(H,21,24)(H,22,25)(H4,18,19,20)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131987
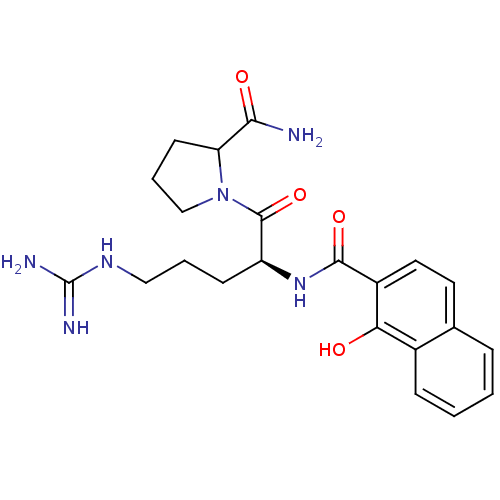
(1-{5-Guanidino-2-[(1-hydroxy-naphthalene-2-carbony...)Show SMILES NC(=N)NCCC[C@H](NC(=O)c1ccc2ccccc2c1O)C(=O)N1CCCC1C(N)=O Show InChI InChI=1S/C22H28N6O4/c23-19(30)17-8-4-12-28(17)21(32)16(7-3-11-26-22(24)25)27-20(31)15-10-9-13-5-1-2-6-14(13)18(15)29/h1-2,5-6,9-10,16-17,29H,3-4,7-8,11-12H2,(H2,23,30)(H,27,31)(H4,24,25,26)/t16-,17?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131977

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,12,14-15,28H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131977

(1-Acetyl-4-hydroxy-pyrrolidine-2-carboxylic acid [...)Show SMILES CC(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O4S/c1-11(27)26-10-12(28)9-15(26)18(30)24-14(6-4-8-23-20(21)22)17(29)19-25-13-5-2-3-7-16(13)31-19/h2-3,5,7,12,14-15,28H,4,6,8-10H2,1H3,(H,24,30)(H4,21,22,23)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131981

(CHEMBL340098 | N-{[1-(Benzothiazole-2-carbonyl)-4-...)Show SMILES CC(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C17H22N6O3S/c1-10(24)21-9-14(25)22-12(6-4-8-20-17(18)19)15(26)16-23-11-5-2-3-7-13(11)27-16/h2-3,5,7,12H,4,6,8-9H2,1H3,(H,21,24)(H,22,25)(H4,18,19,20)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50131976
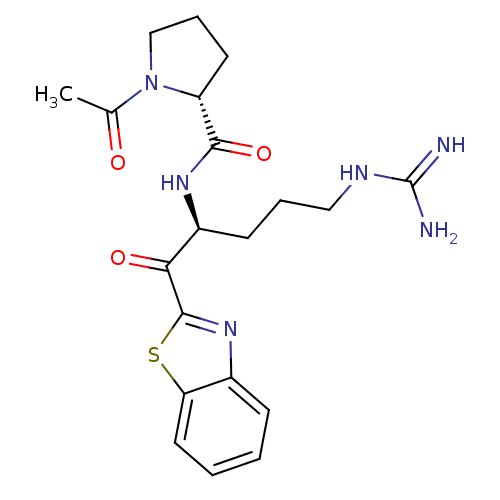
(1-Acetyl-pyrrolidine-2-carboxylic acid [1-(benzoth...)Show SMILES CC(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O3S/c1-12(27)26-11-5-8-15(26)18(29)24-14(7-4-10-23-20(21)22)17(28)19-25-13-6-2-3-9-16(13)30-19/h2-3,6,9,14-15H,4-5,7-8,10-11H2,1H3,(H,24,29)(H4,21,22,23)/t14-,15+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50131976

(1-Acetyl-pyrrolidine-2-carboxylic acid [1-(benzoth...)Show SMILES CC(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)c1nc2ccccc2s1 Show InChI InChI=1S/C20H26N6O3S/c1-12(27)26-11-5-8-15(26)18(29)24-14(7-4-10-23-20(21)22)17(28)19-25-13-6-2-3-9-16(13)30-19/h2-3,6,9,14-15H,4-5,7-8,10-11H2,1H3,(H,24,29)(H4,21,22,23)/t14-,15+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Tryptase beta. |
J Med Chem 46: 3865-76 (2003)
Article DOI: 10.1021/jm030050p
BindingDB Entry DOI: 10.7270/Q23T9J0V |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data