 Found 40 hits with Last Name = 'cyr' and Initial = 'd'
Found 40 hits with Last Name = 'cyr' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300426
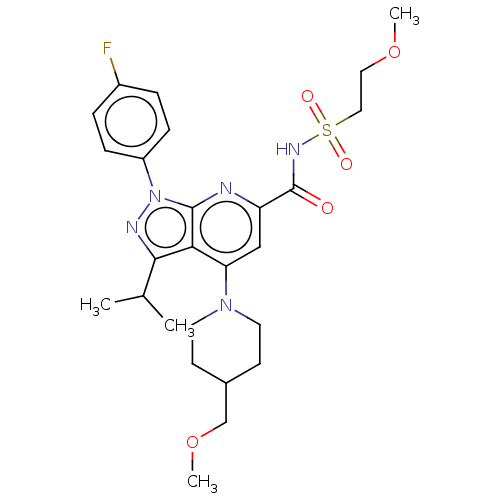
(US10130622, Example 00005)Show SMILES COCCS(=O)(=O)NC(=O)c1cc(N2CCC(COC)CC2)c2c(nn(-c3ccc(F)cc3)c2n1)C(C)C Show InChI InChI=1S/C26H34FN5O5S/c1-17(2)24-23-22(31-11-9-18(10-12-31)16-37-4)15-21(26(33)30-38(34,35)14-13-36-3)28-25(23)32(29-24)20-7-5-19(27)6-8-20/h5-8,15,17-18H,9-14,16H2,1-4H3,(H,30,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
For this purpose, HEK293 cells are transfected with plasmid DNA containing WT CFTR and seeded in 96 well plates (70,000 HEK cells/well). Two days aft... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator [G550E]
(Homo sapiens (Human)) | BDBM300426

(US10130622, Example 00005)Show SMILES COCCS(=O)(=O)NC(=O)c1cc(N2CCC(COC)CC2)c2c(nn(-c3ccc(F)cc3)c2n1)C(C)C Show InChI InChI=1S/C26H34FN5O5S/c1-17(2)24-23-22(31-11-9-18(10-12-31)16-37-4)15-21(26(33)30-38(34,35)14-13-36-3)28-25(23)32(29-24)20-7-5-19(27)6-8-20/h5-8,15,17-18H,9-14,16H2,1-4H3,(H,30,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 102 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
YFP-Halide Influx Assay for the CFTR-ΔF508 Mutation and Suppressor mutants (I539T or G550E)The YFP halide influx assay measures the functionalit... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300426

(US10130622, Example 00005)Show SMILES COCCS(=O)(=O)NC(=O)c1cc(N2CCC(COC)CC2)c2c(nn(-c3ccc(F)cc3)c2n1)C(C)C Show InChI InChI=1S/C26H34FN5O5S/c1-17(2)24-23-22(31-11-9-18(10-12-31)16-37-4)15-21(26(33)30-38(34,35)14-13-36-3)28-25(23)32(29-24)20-7-5-19(27)6-8-20/h5-8,15,17-18H,9-14,16H2,1-4H3,(H,30,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 103 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
For this purpose, HEK293 cells are transfected with plasmid DNA containing WT CFTR and seeded in 96 well plates (70,000 HEK cells/well). Two days aft... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300427
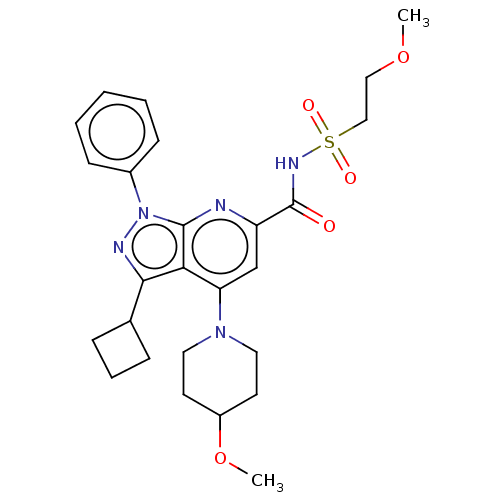
(US10130622, Example 00006)Show SMILES COCCS(=O)(=O)NC(=O)c1cc(N2CCC(CC2)OC)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C26H33N5O5S/c1-35-15-16-37(33,34)29-26(32)21-17-22(30-13-11-20(36-2)12-14-30)23-24(18-7-6-8-18)28-31(25(23)27-21)19-9-4-3-5-10-19/h3-5,9-10,17-18,20H,6-8,11-16H2,1-2H3,(H,29,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 104 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
For this purpose, HEK293 cells are transfected with plasmid DNA containing WT CFTR and seeded in 96 well plates (70,000 HEK cells/well). Two days aft... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300428
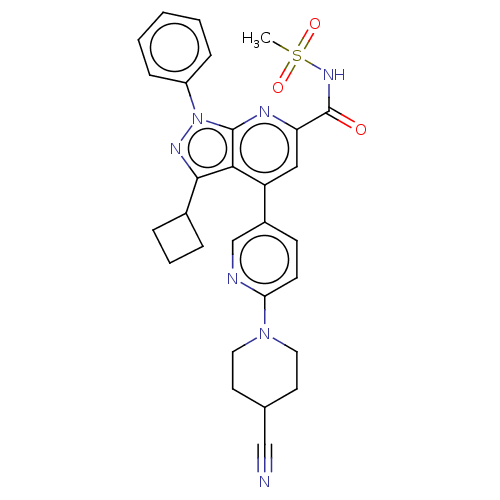
(BDBM300430 | US10130622, Example 00007)Show SMILES CS(=O)(=O)NC(=O)c1cc(-c2ccc(nc2)N2CCC(CC2)C#N)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C29H29N7O3S/c1-40(38,39)34-29(37)24-16-23(21-10-11-25(31-18-21)35-14-12-19(17-30)13-15-35)26-27(20-6-5-7-20)33-36(28(26)32-24)22-8-3-2-4-9-22/h2-4,8-11,16,18-20H,5-7,12-15H2,1H3,(H,34,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 138 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
For this purpose, HEK293 cells are transfected with plasmid DNA containing WT CFTR and seeded in 96 well plates (70,000 HEK cells/well). Two days aft... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator [G550E]
(Homo sapiens (Human)) | BDBM300427

(US10130622, Example 00006)Show SMILES COCCS(=O)(=O)NC(=O)c1cc(N2CCC(CC2)OC)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C26H33N5O5S/c1-35-15-16-37(33,34)29-26(32)21-17-22(30-13-11-20(36-2)12-14-30)23-24(18-7-6-8-18)28-31(25(23)27-21)19-9-4-3-5-10-19/h3-5,9-10,17-18,20H,6-8,11-16H2,1-2H3,(H,29,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 417 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
YFP-Halide Influx Assay for the CFTR-ΔF508 Mutation and Suppressor mutants (I539T or G550E)The YFP halide influx assay measures the functionalit... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator [G550E]
(Homo sapiens (Human)) | BDBM300428

(BDBM300430 | US10130622, Example 00007)Show SMILES CS(=O)(=O)NC(=O)c1cc(-c2ccc(nc2)N2CCC(CC2)C#N)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C29H29N7O3S/c1-40(38,39)34-29(37)24-16-23(21-10-11-25(31-18-21)35-14-12-19(17-30)13-15-35)26-27(20-6-5-7-20)33-36(28(26)32-24)22-8-3-2-4-9-22/h2-4,8-11,16,18-20H,5-7,12-15H2,1H3,(H,34,37) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 496 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
YFP-Halide Influx Assay for the CFTR-ΔF508 Mutation and Suppressor mutants (I539T or G550E)The YFP halide influx assay measures the functionalit... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300428

(BDBM300430 | US10130622, Example 00007)Show SMILES CS(=O)(=O)NC(=O)c1cc(-c2ccc(nc2)N2CCC(CC2)C#N)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C29H29N7O3S/c1-40(38,39)34-29(37)24-16-23(21-10-11-25(31-18-21)35-14-12-19(17-30)13-15-35)26-27(20-6-5-7-20)33-36(28(26)32-24)22-8-3-2-4-9-22/h2-4,8-11,16,18-20H,5-7,12-15H2,1H3,(H,34,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
For this purpose, HEK293 cells are transfected with plasmid DNA containing WT CFTR and seeded in 96 well plates (70,000 HEK cells/well). Two days aft... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589806

(CHEMBL5177469)Show SMILES O=C(CNCc1cc2ccccc2[nH]1)N[C@H]1CC[C@H](CC1)c1nc2ccccc2[nH]1 |r,wU:15.16,18.23,(-1.11,.83,;-1.88,2.16,;-3.42,2.16,;-4.19,.83,;-5.73,.83,;-6.5,-.51,;-6.03,-1.97,;-7.27,-2.88,;-7.43,-4.4,;-8.84,-5.03,;-10.08,-4.13,;-9.93,-2.6,;-8.52,-1.97,;-8.04,-.51,;-1.11,3.49,;.43,3.49,;1.2,4.83,;2.74,4.83,;3.51,3.49,;2.74,2.16,;1.2,2.16,;5.05,3.49,;5.95,2.25,;7.42,2.72,;8.76,1.95,;10.08,2.73,;10.08,4.26,;8.75,5.03,;7.42,4.26,;5.95,4.74,)| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589802

(CHEMBL5177515)Show SMILES CCCCNCC(=O)N[C@H](Cc1ccc(cc1)C(C)(C)C)C(=O)N[C@@H](Cc1cccc(OC)c1)C(=O)NC |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589804

(CHEMBL5191076)Show SMILES CNC(=O)[C@H](Cc1ccc(C)cc1)NC(=O)[C@@H](C\C=C\c1ccccc1)NC(=O)CNCc1ccccc1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589801
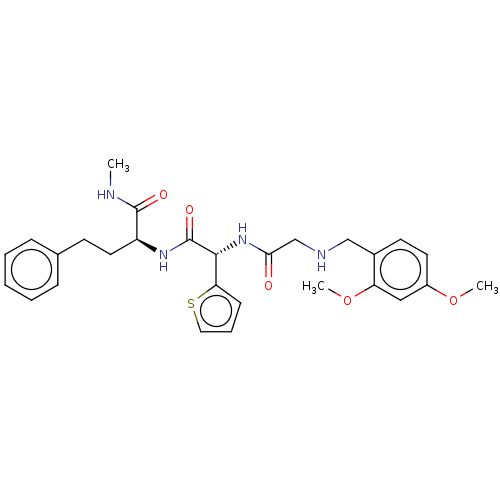
(CHEMBL5208789)Show SMILES CNC(=O)[C@H](CCc1ccccc1)NC(=O)[C@H](NC(=O)CNCc1ccc(OC)cc1OC)c1cccs1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300427

(US10130622, Example 00006)Show SMILES COCCS(=O)(=O)NC(=O)c1cc(N2CCC(CC2)OC)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C26H33N5O5S/c1-35-15-16-37(33,34)29-26(32)21-17-22(30-13-11-20(36-2)12-14-30)23-24(18-7-6-8-18)28-31(25(23)27-21)19-9-4-3-5-10-19/h3-5,9-10,17-18,20H,6-8,11-16H2,1-2H3,(H,29,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | >6.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
For this purpose, HEK293 cells are transfected with plasmid DNA containing WT CFTR and seeded in 96 well plates (70,000 HEK cells/well). Two days aft... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589797
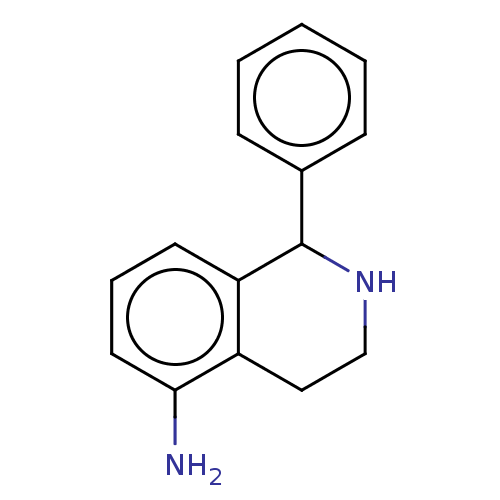
(CHEMBL5189044) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 1.89E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589800

(CHEMBL5197897)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.14E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589799
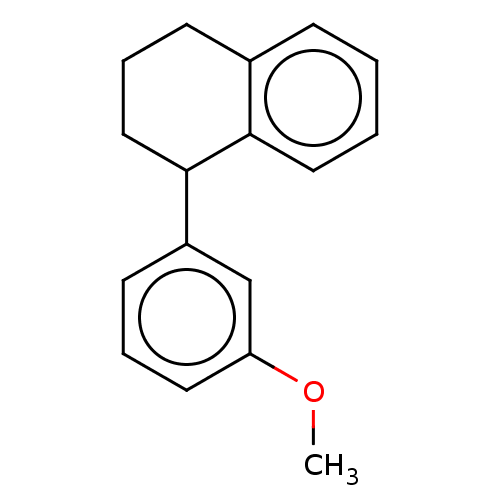
(CHEMBL5175764) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.55E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589791

(CHEMBL5179578) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.93E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589790

(CHEMBL5194502) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589796

(CHEMBL5193368) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.48E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50017043
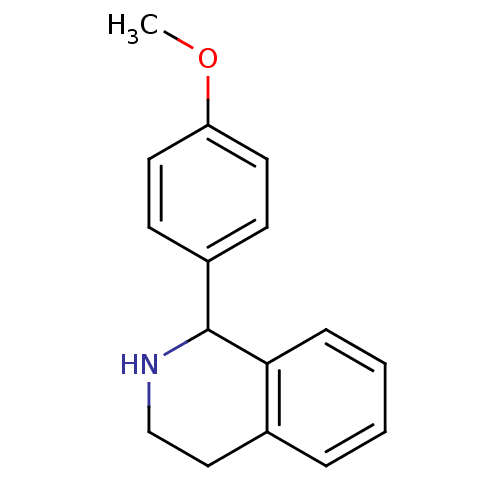
(1-(4-Methoxy-phenyl)-1,2,3,4-tetrahydro-isoquinoli...)Show InChI InChI=1S/C16H17NO/c1-18-14-8-6-13(7-9-14)16-15-5-3-2-4-12(15)10-11-17-16/h2-9,16-17H,10-11H2,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.98E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589787

(CHEMBL5179037) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.08E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589788
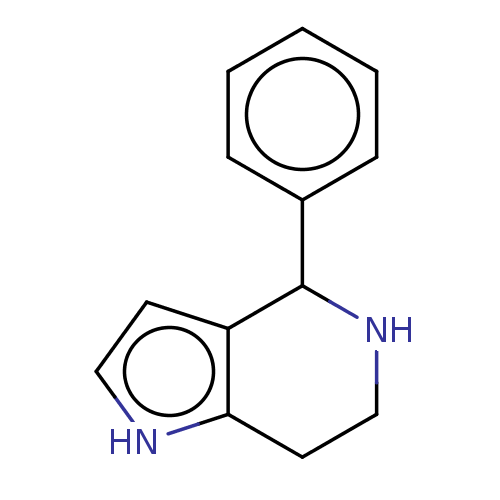
(CHEMBL5188512) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.08E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589785
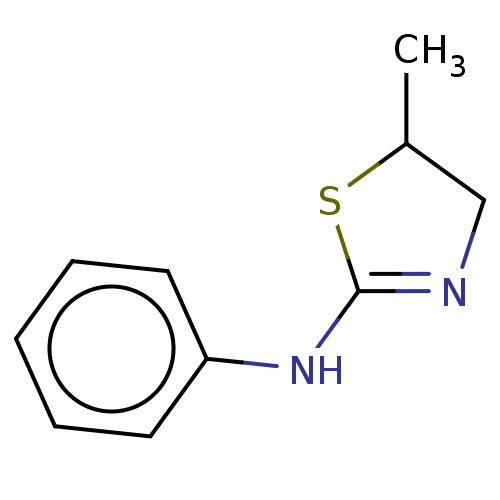
(CHEMBL1600521) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.13E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589784
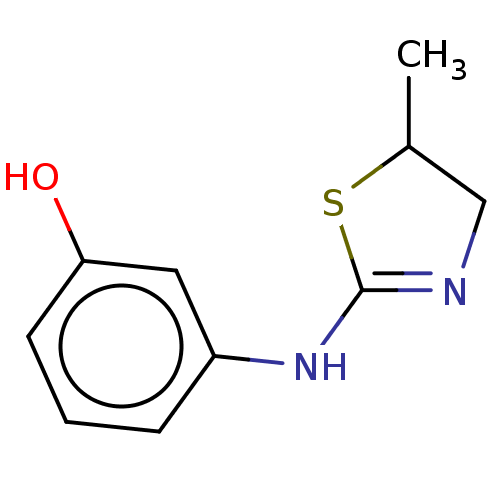
(CHEMBL5186109) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 1.49E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589795

(CHEMBL5195783) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.49E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589803

(CHEMBL5208155)Show SMILES CNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@@H](NC(=O)[C@H]1NC[C@@H]1c1ccccc1)c1ccsc1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.88E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589805

(CHEMBL5191415)Show SMILES CNC(=O)C1N(Cc2ccccc12)C(=O)[C@@H](Cc1c(C)[nH]c2ccccc12)NC(=O)CNCCOc1ccc(C)cc1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.88E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589793
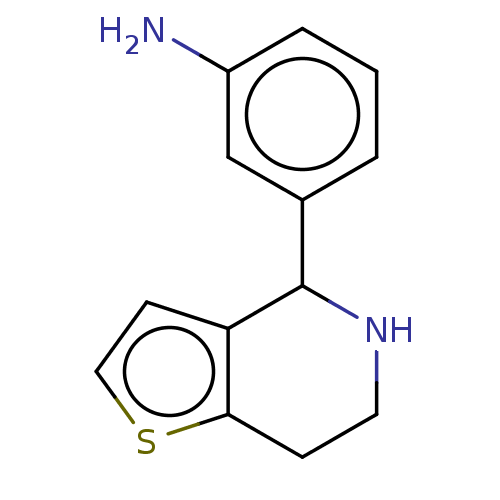
(CHEMBL5198959) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.35E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589798
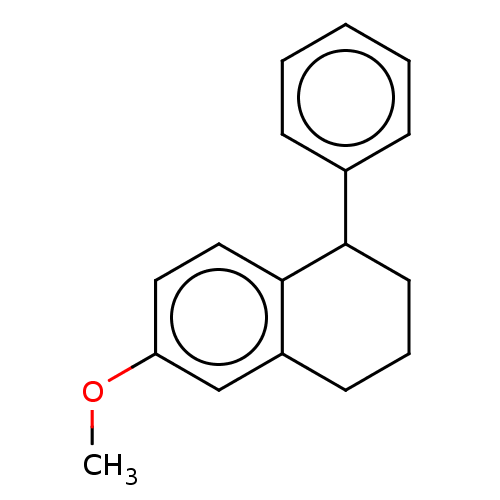
(CHEMBL5181147) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589786
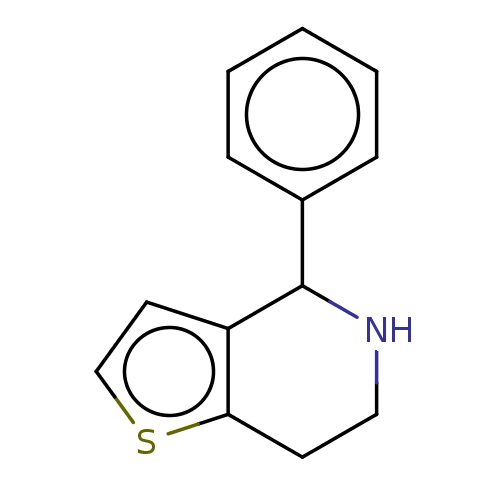
(CHEMBL2179396) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589794

(CHEMBL5203207) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589792
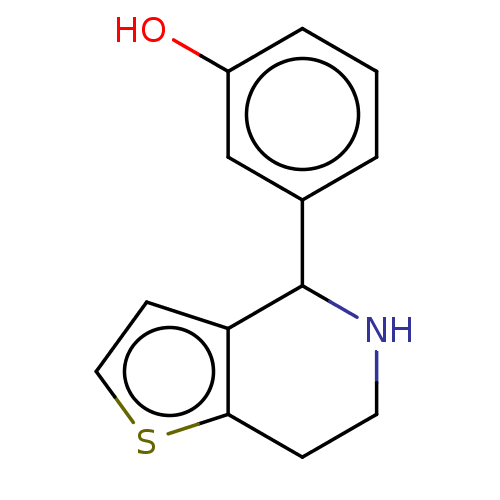
(CHEMBL5205965) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589789

(CHEMBL5192951) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589783

(CHEMBL1998455) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.64E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589784

(CHEMBL5186109) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | 1.10E+5 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589797

(CHEMBL5189044) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300429
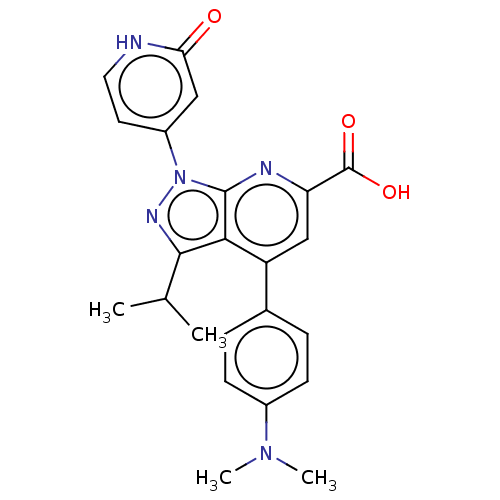
(US10130622, Example 00008)Show SMILES CC(C)c1nn(-c2cc[nH]c(=O)c2)c2nc(cc(-c3ccc(cc3)N(C)C)c12)C(O)=O Show InChI InChI=1S/C23H23N5O3/c1-13(2)21-20-17(14-5-7-15(8-6-14)27(3)4)12-18(23(30)31)25-22(20)28(26-21)16-9-10-24-19(29)11-16/h5-13H,1-4H3,(H,24,29)(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | 9.66E+3 | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
To measure the direct binding of small molecules with CFTR, the interaction between small molecules and membrane fragments derived from HEK293 cells ... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Cystic fibrosis transmembrane conductance regulator
(Homo sapiens (Human)) | BDBM300428

(BDBM300430 | US10130622, Example 00007)Show SMILES CS(=O)(=O)NC(=O)c1cc(-c2ccc(nc2)N2CCC(CC2)C#N)c2c(nn(-c3ccccc3)c2n1)C1CCC1 Show InChI InChI=1S/C29H29N7O3S/c1-40(38,39)34-29(37)24-16-23(21-10-11-25(31-18-21)35-14-12-19(17-30)13-15-35)26-27(20-6-5-7-20)33-36(28(26)32-24)22-8-3-2-4-9-22/h2-4,8-11,16,18-20H,5-7,12-15H2,1H3,(H,34,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | 20.2 | n/a | n/a | n/a | n/a | n/a |
AbbVie S.á.r.l.; Galapagos NV
US Patent
| Assay Description
To measure the direct binding of small molecules with CFTR, the interaction between small molecules and membrane fragments derived from HEK293 cells ... |
US Patent US10130622 (2018)
BindingDB Entry DOI: 10.7270/Q2KD20ZB |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589782
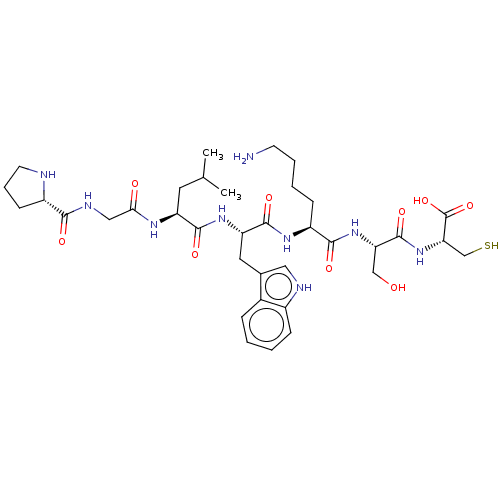
(CHEMBL5195622)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | |
Glucose-induced degradation protein 4 homolog
(Homo sapiens) | BDBM50589806

(CHEMBL5177469)Show SMILES O=C(CNCc1cc2ccccc2[nH]1)N[C@H]1CC[C@H](CC1)c1nc2ccccc2[nH]1 |r,wU:15.16,18.23,(-1.11,.83,;-1.88,2.16,;-3.42,2.16,;-4.19,.83,;-5.73,.83,;-6.5,-.51,;-6.03,-1.97,;-7.27,-2.88,;-7.43,-4.4,;-8.84,-5.03,;-10.08,-4.13,;-9.93,-2.6,;-8.52,-1.97,;-8.04,-.51,;-1.11,3.49,;.43,3.49,;1.2,4.83,;2.74,4.83,;3.51,3.49,;2.74,2.16,;1.2,2.16,;5.05,3.49,;5.95,2.25,;7.42,2.72,;8.76,1.95,;10.08,2.73,;10.08,4.26,;8.75,5.03,;7.42,4.26,;5.95,4.74,)| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00509
BindingDB Entry DOI: 10.7270/Q2JD51RW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data