 Found 77 hits with Last Name = 'datta' and Initial = 'd'
Found 77 hits with Last Name = 'datta' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438455
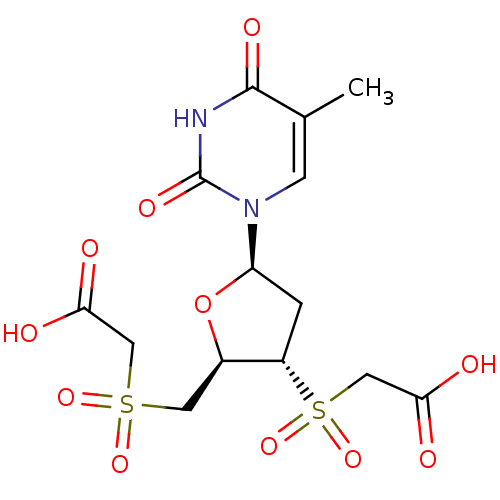
(CHEMBL2414522)Show SMILES Cc1cn([C@H]2C[C@@H]([C@@H](CS(=O)(=O)CC(O)=O)O2)S(=O)(=O)CC(O)=O)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C14H18N2O11S2/c1-7-3-16(14(22)15-13(7)21)10-2-9(29(25,26)6-12(19)20)8(27-10)4-28(23,24)5-11(17)18/h3,8-10H,2,4-6H2,1H3,(H,17,18)(H,19,20)(H,15,21,22)/t8-,9+,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438453
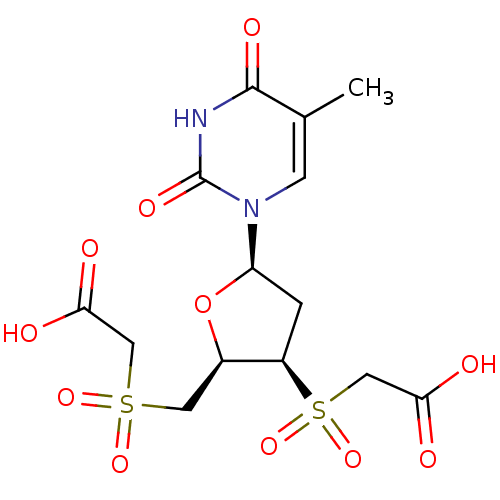
(CHEMBL2414524)Show SMILES Cc1cn([C@H]2C[C@H]([C@@H](CS(=O)(=O)CC(O)=O)O2)S(=O)(=O)CC(O)=O)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C14H18N2O11S2/c1-7-3-16(14(22)15-13(7)21)10-2-9(29(25,26)6-12(19)20)8(27-10)4-28(23,24)5-11(17)18/h3,8-10H,2,4-6H2,1H3,(H,17,18)(H,19,20)(H,15,21,22)/t8-,9-,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438456
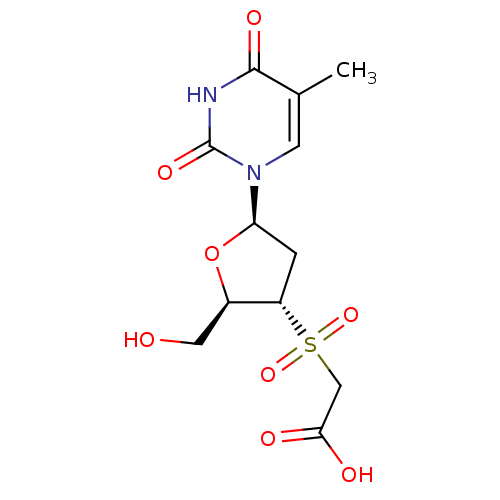
(CHEMBL2414521)Show SMILES Cc1cn([C@H]2C[C@@H]([C@@H](CO)O2)S(=O)(=O)CC(O)=O)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C12H16N2O8S/c1-6-3-14(12(19)13-11(6)18)9-2-8(7(4-15)22-9)23(20,21)5-10(16)17/h3,7-9,15H,2,4-5H2,1H3,(H,16,17)(H,13,18,19)/t7-,8+,9-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438454
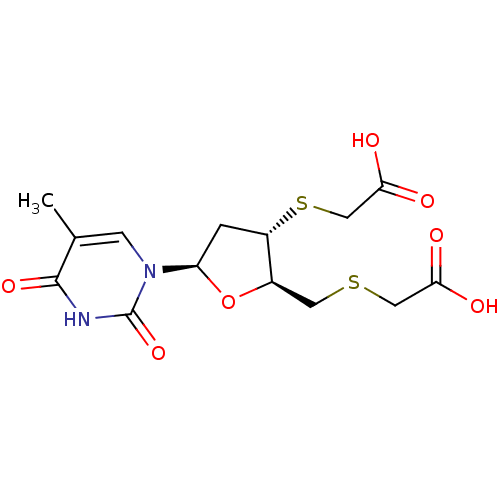
(CHEMBL2414523)Show SMILES Cc1cn([C@H]2C[C@H](SCC(O)=O)[C@@H](CSCC(O)=O)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C14H18N2O7S2/c1-7-3-16(14(22)15-13(7)21)10-2-9(25-6-12(19)20)8(23-10)4-24-5-11(17)18/h3,8-10H,2,4-6H2,1H3,(H,17,18)(H,19,20)(H,15,21,22)/t8-,9+,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438451

(CHEMBL2414510)Show SMILES Cc1cn([C@H]2C[C@H](O)[C@@H](CS(=O)(=O)CC(O)=O)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C12H16N2O8S/c1-6-3-14(12(19)13-11(6)18)9-2-7(15)8(22-9)4-23(20,21)5-10(16)17/h3,7-9,15H,2,4-5H2,1H3,(H,16,17)(H,13,18,19)/t7-,8+,9+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438452
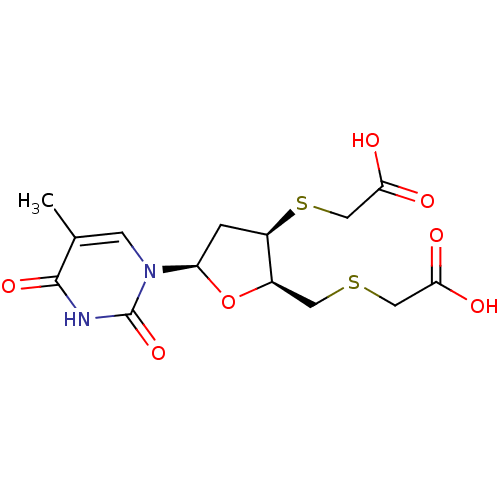
(CHEMBL2414509)Show SMILES Cc1cn([C@H]2C[C@@H](SCC(O)=O)[C@@H](CSCC(O)=O)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C14H18N2O7S2/c1-7-3-16(14(22)15-13(7)21)10-2-9(25-6-12(19)20)8(23-10)4-24-5-11(17)18/h3,8-10H,2,4-6H2,1H3,(H,17,18)(H,19,20)(H,15,21,22)/t8-,9-,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.08E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438448
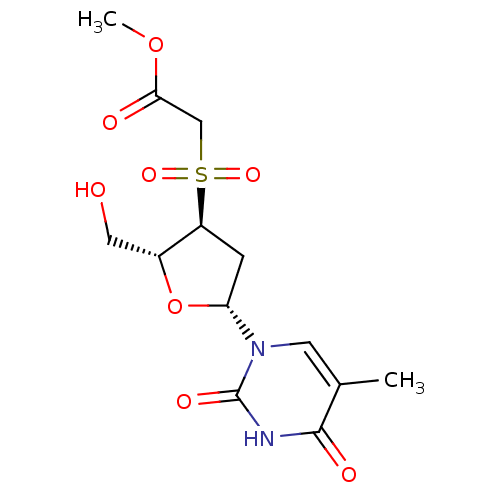
(CHEMBL2414513)Show SMILES COC(=O)CS(=O)(=O)[C@H]1C[C@@H](O[C@@H]1CO)n1cc(C)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C13H18N2O8S/c1-7-4-15(13(19)14-12(7)18)10-3-9(8(5-16)23-10)24(20,21)6-11(17)22-2/h4,8-10,16H,3,5-6H2,1-2H3,(H,14,18,19)/t8-,9+,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438457
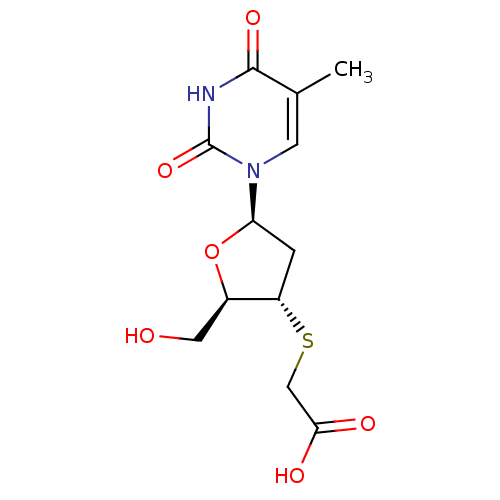
(CHEMBL2414520)Show SMILES Cc1cn([C@H]2C[C@H](SCC(O)=O)[C@@H](CO)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C12H16N2O6S/c1-6-3-14(12(19)13-11(6)18)9-2-8(7(4-15)20-9)21-5-10(16)17/h3,7-9,15H,2,4-5H2,1H3,(H,16,17)(H,13,18,19)/t7-,8+,9-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.69E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438458

(CHEMBL2414519)Show SMILES Cc1cn([C@H]2C[C@H](NCC(O)=O)[C@@H](CO)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C12H17N3O6/c1-6-4-15(12(20)14-11(6)19)9-2-7(8(5-16)21-9)13-3-10(17)18/h4,7-9,13,16H,2-3,5H2,1H3,(H,17,18)(H,14,19,20)/t7-,8+,9+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.86E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438445
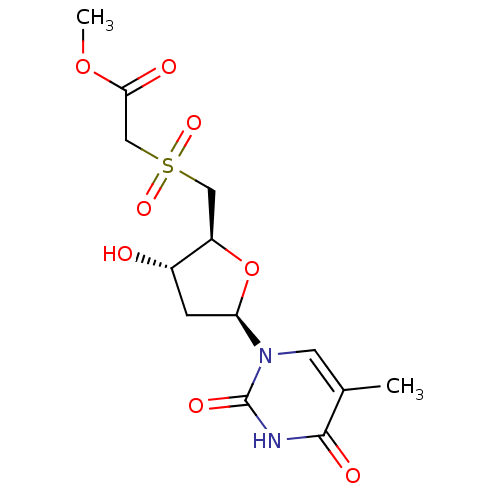
(CHEMBL2414517)Show SMILES COC(=O)CS(=O)(=O)C[C@H]1O[C@H](C[C@@H]1O)n1cc(C)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C13H18N2O8S/c1-7-4-15(13(19)14-12(7)18)10-3-8(16)9(23-10)5-24(20,21)6-11(17)22-2/h4,8-10,16H,3,5-6H2,1-2H3,(H,14,18,19)/t8-,9+,10+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.02E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438446
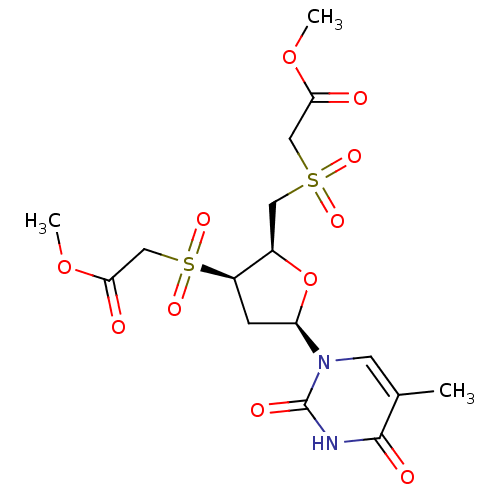
(CHEMBL2414515)Show SMILES COC(=O)CS(=O)(=O)C[C@H]1O[C@H](C[C@H]1S(=O)(=O)CC(=O)OC)n1cc(C)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C16H22N2O11S2/c1-9-5-18(16(22)17-15(9)21)12-4-11(31(25,26)8-14(20)28-3)10(29-12)6-30(23,24)7-13(19)27-2/h5,10-12H,4,6-8H2,1-3H3,(H,17,21,22)/t10-,11-,12-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.42E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438447
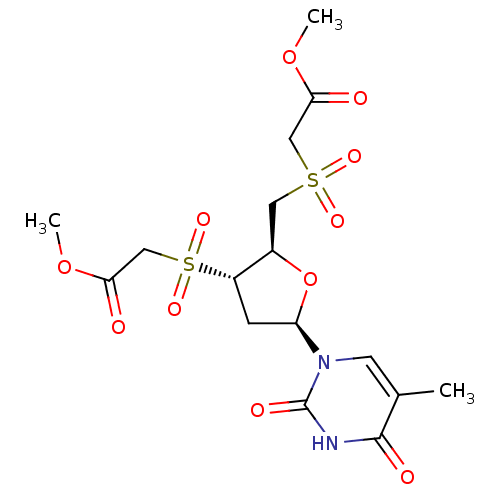
(CHEMBL2414514)Show SMILES COC(=O)CS(=O)(=O)C[C@H]1O[C@H](C[C@@H]1S(=O)(=O)CC(=O)OC)n1cc(C)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C16H22N2O11S2/c1-9-5-18(16(22)17-15(9)21)12-4-11(31(25,26)8-14(20)28-3)10(29-12)6-30(23,24)7-13(19)27-2/h5,10-12H,4,6-8H2,1-3H3,(H,17,21,22)/t10-,11+,12-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.67E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438459
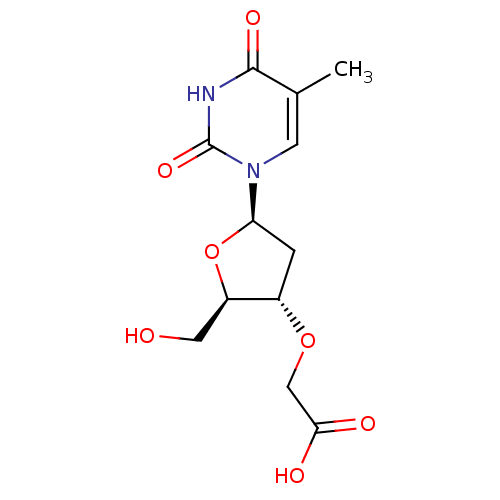
(CHEMBL2414518)Show SMILES Cc1cn([C@H]2C[C@H](OCC(O)=O)[C@@H](CO)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C12H16N2O7/c1-6-3-14(12(19)13-11(6)18)9-2-7(8(4-15)21-9)20-5-10(16)17/h3,7-9,15H,2,4-5H2,1H3,(H,16,17)(H,13,18,19)/t7-,8+,9+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.67E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438450
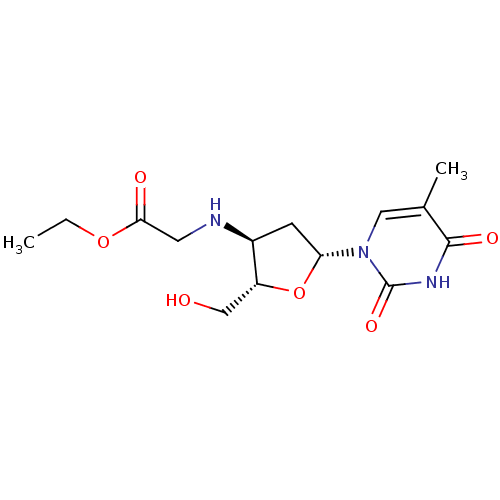
(CHEMBL2414511)Show SMILES CCOC(=O)CN[C@H]1C[C@@H](O[C@@H]1CO)n1cc(C)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C14H21N3O6/c1-3-22-12(19)5-15-9-4-11(23-10(9)7-18)17-6-8(2)13(20)16-14(17)21/h6,9-11,15,18H,3-5,7H2,1-2H3,(H,16,20,21)/t9-,10+,11+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.04E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Ribonuclease pancreatic
(Bison bison (American bison)) | BDBM50438449
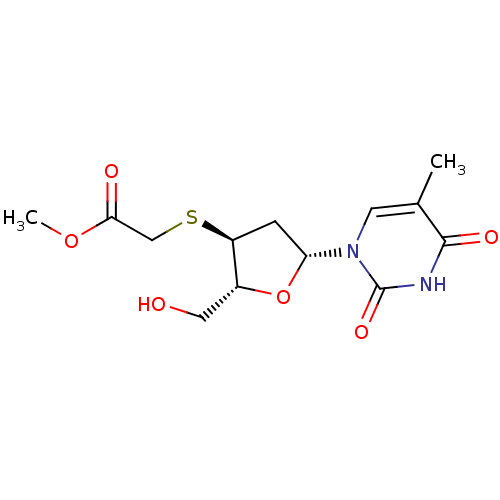
(CHEMBL2414512)Show SMILES COC(=O)CS[C@H]1C[C@@H](O[C@@H]1CO)n1cc(C)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C13H18N2O6S/c1-7-4-15(13(19)14-12(7)18)10-3-9(8(5-16)21-10)22-6-11(17)20-2/h4,8-10,16H,3,5-6H2,1-2H3,(H,14,18,19)/t8-,9+,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.57E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Technology Kharagpur
Curated by ChEMBL
| Assay Description
Reversible competitive inhibition of bovine pancreatic RNase A using 2',3'-cCMP as substrate by Lineweaver-Burk plot analysis |
Bioorg Med Chem 21: 4634-45 (2013)
Article DOI: 10.1016/j.bmc.2013.05.047
BindingDB Entry DOI: 10.7270/Q2WQ056P |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50367254

(ENALAPRILAT)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1CCC[C@H]1C(O)=O |r| Show InChI InChI=1S/C18H24N2O5/c1-12(16(21)20-11-5-8-15(20)18(24)25)19-14(17(22)23)10-9-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,19H,5,8-11H2,1H3,(H,22,23)(H,24,25)/t12-,14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Rattus norvegicus) | BDBM50367254

(ENALAPRILAT)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1CCC[C@H]1C(O)=O |r| Show InChI InChI=1S/C18H24N2O5/c1-12(16(21)20-11-5-8-15(20)18(24)25)19-14(17(22)23)10-9-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,19H,5,8-11H2,1H3,(H,22,23)(H,24,25)/t12-,14-,15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Mus musculus) | BDBM50367254

(ENALAPRILAT)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1CCC[C@H]1C(O)=O |r| Show InChI InChI=1S/C18H24N2O5/c1-12(16(21)20-11-5-8-15(20)18(24)25)19-14(17(22)23)10-9-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,19H,5,8-11H2,1H3,(H,22,23)(H,24,25)/t12-,14-,15-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11162
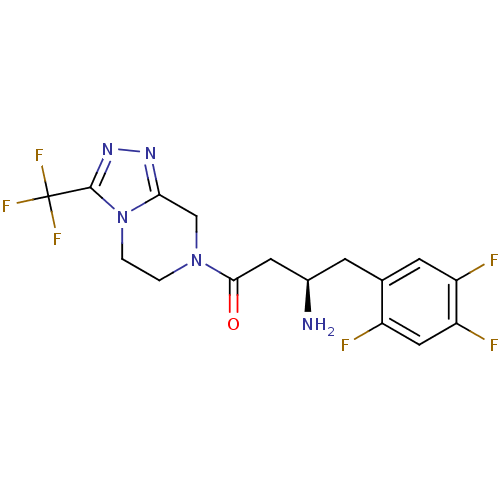
((1R)-3-oxo-3-[3-(trifluoroethyl)-5,6-dihydro[1,2,4...)Show SMILES N[C@@H](CC(=O)N1CCn2c(C1)nnc2C(F)(F)F)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C16H15F6N5O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22/h4,6,9H,1-3,5,7,23H2/t9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiotensin-converting enzyme
(Rattus norvegicus) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Rattus norvegicus (rat)) | BDBM11162

((1R)-3-oxo-3-[3-(trifluoroethyl)-5,6-dihydro[1,2,4...)Show SMILES N[C@@H](CC(=O)N1CCn2c(C1)nnc2C(F)(F)F)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C16H15F6N5O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22/h4,6,9H,1-3,5,7,23H2/t9-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Compound was tested in vitro for binding affinity against human carbonic anhydrase II; (ki*10e-9) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Mus musculus) | BDBM11162

((1R)-3-oxo-3-[3-(trifluoroethyl)-5,6-dihydro[1,2,4...)Show SMILES N[C@@H](CC(=O)N1CCn2c(C1)nnc2C(F)(F)F)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C16H15F6N5O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22/h4,6,9H,1-3,5,7,23H2/t9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242265
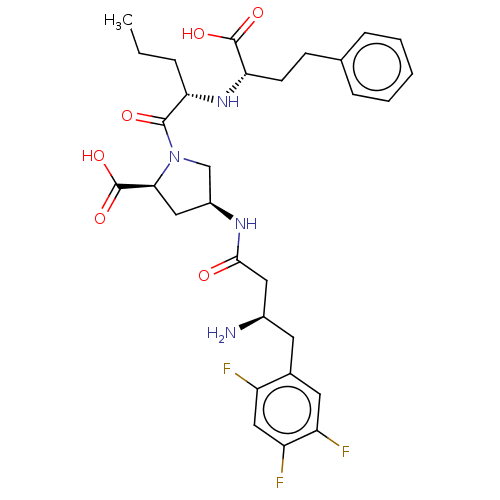
(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 57 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Mus musculus) | BDBM50242266
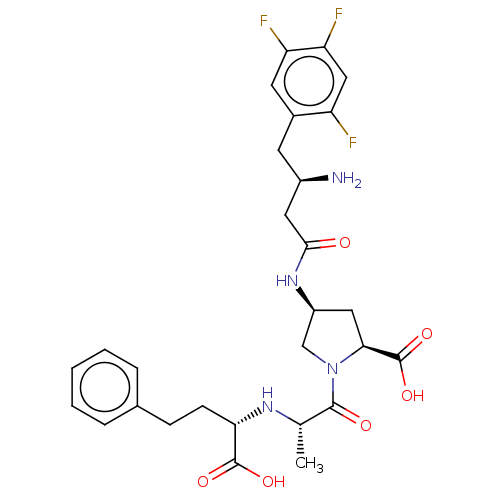
(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 58 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50242265

(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Mus musculus) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50242274
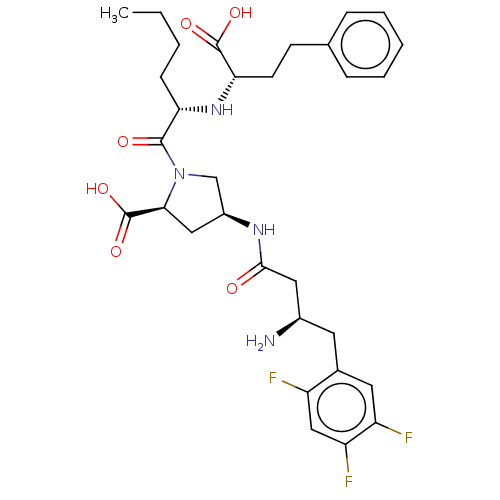
(CHEMBL4090635)Show SMILES CCCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C31H39F3N4O6/c1-2-3-9-25(37-26(30(41)42)11-10-18-7-5-4-6-8-18)29(40)38-17-21(15-27(38)31(43)44)36-28(39)14-20(35)12-19-13-23(33)24(34)16-22(19)32/h4-8,13,16,20-21,25-27,37H,2-3,9-12,14-15,17,35H2,1H3,(H,36,39)(H,41,42)(H,43,44)/t20-,21+,25+,26+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 102 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Rattus norvegicus (rat)) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 155 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 158 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Mus musculus) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 217 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 255 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242274

(CHEMBL4090635)Show SMILES CCCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C31H39F3N4O6/c1-2-3-9-25(37-26(30(41)42)11-10-18-7-5-4-6-8-18)29(40)38-17-21(15-27(38)31(43)44)36-28(39)14-20(35)12-19-13-23(33)24(34)16-22(19)32/h4-8,13,16,20-21,25-27,37H,2-3,9-12,14-15,17,35H2,1H3,(H,36,39)(H,41,42)(H,43,44)/t20-,21+,25+,26+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 279 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Rattus norvegicus (rat)) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 319 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Rattus norvegicus) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 334 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Mus musculus) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 488 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Rattus norvegicus) | BDBM50242265

(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 893 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Rattus norvegicus (rat)) | BDBM50242265

(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.23E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Mus musculus) | BDBM50242265

(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Mus musculus) | BDBM50242274

(CHEMBL4090635)Show SMILES CCCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C31H39F3N4O6/c1-2-3-9-25(37-26(30(41)42)11-10-18-7-5-4-6-8-18)29(40)38-17-21(15-27(38)31(43)44)36-28(39)14-20(35)12-19-13-23(33)24(34)16-22(19)32/h4-8,13,16,20-21,25-27,37H,2-3,9-12,14-15,17,35H2,1H3,(H,36,39)(H,41,42)(H,43,44)/t20-,21+,25+,26+,27+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.21E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Mus musculus) | BDBM50242265

(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Rattus norvegicus) | BDBM50242274

(CHEMBL4090635)Show SMILES CCCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C31H39F3N4O6/c1-2-3-9-25(37-26(30(41)42)11-10-18-7-5-4-6-8-18)29(40)38-17-21(15-27(38)31(43)44)36-28(39)14-20(35)12-19-13-23(33)24(34)16-22(19)32/h4-8,13,16,20-21,25-27,37H,2-3,9-12,14-15,17,35H2,1H3,(H,36,39)(H,41,42)(H,43,44)/t20-,21+,25+,26+,27+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Rattus norvegicus (rat)) | BDBM50242274

(CHEMBL4090635)Show SMILES CCCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C31H39F3N4O6/c1-2-3-9-25(37-26(30(41)42)11-10-18-7-5-4-6-8-18)29(40)38-17-21(15-27(38)31(43)44)36-28(39)14-20(35)12-19-13-23(33)24(34)16-22(19)32/h4-8,13,16,20-21,25-27,37H,2-3,9-12,14-15,17,35H2,1H3,(H,36,39)(H,41,42)(H,43,44)/t20-,21+,25+,26+,27+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Mus musculus) | BDBM50242274

(CHEMBL4090635)Show SMILES CCCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C31H39F3N4O6/c1-2-3-9-25(37-26(30(41)42)11-10-18-7-5-4-6-8-18)29(40)38-17-21(15-27(38)31(43)44)36-28(39)14-20(35)12-19-13-23(33)24(34)16-22(19)32/h4-8,13,16,20-21,25-27,37H,2-3,9-12,14-15,17,35H2,1H3,(H,36,39)(H,41,42)(H,43,44)/t20-,21+,25+,26+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of ob/ob mouse plasma DPP4 |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM11162

((1R)-3-oxo-3-[3-(trifluoroethyl)-5,6-dihydro[1,2,4...)Show SMILES N[C@@H](CC(=O)N1CCn2c(C1)nnc2C(F)(F)F)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C16H15F6N5O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22/h4,6,9H,1-3,5,7,23H2/t9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 8
(Homo sapiens (Human)) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of DPP8 (unknown origin) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Prolyl endopeptidase
(Homo sapiens (Human)) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.16E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of PPCE (unknown origin) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 9
(Homo sapiens (Human)) | BDBM50242266

(CHEMBL4067185)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C28H33F3N4O6/c1-15(33-23(27(38)39)8-7-16-5-3-2-4-6-16)26(37)35-14-19(12-24(35)28(40)41)34-25(36)11-18(32)9-17-10-21(30)22(31)13-20(17)29/h2-6,10,13,15,18-19,23-24,33H,7-9,11-12,14,32H2,1H3,(H,34,36)(H,38,39)(H,40,41)/t15-,18+,19-,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Compound was tested in vitro for binding affinity against human carbonic anhydrase II; (ki*10e-9) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 8
(Homo sapiens (Human)) | BDBM50242265

(CHEMBL4078112)Show SMILES CCC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C30H37F3N4O6/c1-2-6-24(36-25(29(40)41)10-9-17-7-4-3-5-8-17)28(39)37-16-20(14-26(37)30(42)43)35-27(38)13-19(34)11-18-12-22(32)23(33)15-21(18)31/h3-5,7-8,12,15,19-20,24-26,36H,2,6,9-11,13-14,16,34H2,1H3,(H,35,38)(H,40,41)(H,42,43)/t19-,20+,24+,25+,26+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of DPP8 (unknown origin) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data